高糖刺激下大鼠腎小球系膜細胞MMP-2,TIMP-2,MT1-MMP和CTGF的表達及意義
姚芳,李志紅,閆喆,劉青娟,段惠軍,郄濤
高糖刺激下大鼠腎小球系膜細胞MMP-2,TIMP-2,MT1-MMP和CTGF的表達及意義
姚芳,李志紅,閆喆,劉青娟,段惠軍,郄濤
目的觀察高糖刺激的大鼠腎小球系膜細胞基質金屬蛋白酶-2(MMP-2)及其組織抑制物-2(TIMP-2)、膜型基質金屬蛋白酶-1(MT1-MMP)和結締組織生長因子(CTGF)的動態變化以探討糖尿病腎病(DN)的發病機制。方法體外培養的大鼠HBZY-1腎小球系膜細胞分為低糖(5.5mmol/L葡萄糖)組、高糖(30mmol/L葡萄糖)組和滲透壓對照(5.5mmol/L葡萄糖+24.5mmol/L甘露醇)組,24、48、72、96h后采用RT-PCR及Western blotting法分別檢測MMP-2、TIMP-2、MT1-MMP及CTGF的mRNA及蛋白表達情況,酶聯免疫吸附法(ELISA)檢測培養上清中Ⅳ型膠原的含量。結果Western blotting 結果顯示,與低糖組相比,高糖組MMP-2的表達在24h時略有升高,較低糖組增加10%±4%(P<0.05),至48h時則較低糖組減少42%±2%,其后隨時間延長表達持續降低,至96h時較低糖組減少78%±2%;MT1-MMP表達在24h開始下降并隨時間呈下降趨勢,與低糖組相比較,在刺激的24h,高糖組MT1-MMP表達下降了29%±3%,隨后持續下降,至96h則下降了78%±9%(P<0.01)。高糖組各時間點TIMP-2和CTGF表達均較低糖組增高,其中CTGF的表達在高糖刺激的24h即顯著增高,為低糖組的201%±24%,隨后持續增高,至培養96h為低糖組的484%±51%(P<0.01);TIMP-2的表達在24h時較低糖組增加55%±3%,且隨時間延長呈增高趨勢(P<0.01)。MMP-2、TIMP-2、MT1-MMP和CTGF的mRNA表達與相應蛋白的表達趨勢基本一致。與低糖組相比,高糖組細胞上清中的Ⅳ型膠原于24h即有增加,且持續增高至96h(P<0.05)。低糖組和滲透壓對照組組內、組間的各指標差異均無統計學意義。結論盡管高糖刺激早期可小幅誘導MMP-2的表達增強,但長期高糖刺激則可抑制MMP-2和MT1-MMP的表達及活化,同時促進系膜細胞TIMP-2和CTGF的表達。DN中腎小球細胞外基質的積聚可能是由于上述細胞因子和蛋白酶引起細胞外基質代謝失衡所致。
糖尿病腎病;腎小球系膜細胞;基質金屬蛋白酶-2;基質金屬蛋白酶組織抑制物-2;結締組織生長因子;膜型基質金屬蛋白酶-1
糖尿病腎病(diabetic nephropathy,DN)是糖尿病最常見而嚴重的并發癥,其發生率呈逐年上升趨勢,已成為發達國家終末期腎病的首要原因[1]。DN的主要病理特征包括腎小球硬化及小管間質纖維化。細胞外基質(extracellular matrix,ECM)的積聚是DN中腎小球硬化的基礎。近年來的研究顯示ECM降解能力下降在DN的發生、發展中起著更為重要的作用[2]。基質金屬蛋白酶(metalloproteinases,MMPs)家族中的MMP-2主要參與Ⅳ型膠原的降解,而后者是DN中腎小球基底膜增厚和系膜基質堆積的主要成分,因此與DN的關系最為密切[3]。近年來有關MMP-2及其組織抑制物(tissue inhibitor of metalloproteinase-2,TIMP-2)在DN中的作用存有較多爭議,關于MMP-2酶原的特異膜型激活劑——膜型基質金屬蛋白酶-1(membrane type 1-matrix metalloproteinase,MT1-MMP)在DN中的表達及作用鮮見報道。結締組織生長因子(connective tissue growth factor,CTGF)是TGF-β1下游的重要效應因子,因其具有較強的致纖維化作用[4],且生物效應較單一,已成為DN研究的熱點。目前有關CTGF在DN的ECM代謝障礙中的作用機制研究報道較少。本實驗以腎小球中功能最活躍的系膜細胞作為研究對象,動態觀察高糖刺激對體外培養的大鼠系膜細胞CTGF、MMP-2,TIMP-2和MT1-MMP及ECM表達的影響,以探討DN中ECM代謝障礙的發生機制。
1 材料與方法
1.1 主要材料及試劑 大鼠HBZY-1腎小球系膜細胞株購自武漢大學保藏中心。羊抗CTGF、TIMP-2、MT1-MMP多克隆抗體,小鼠抗MMP-2、β-actin單克隆抗體,ECL增強化學發光試劑盒為Santa Cruz公司產品。RT-PCR試劑盒為Promega公司產品。Ⅳ型膠原ELISA試劑盒為Sigma公司產品。辣根過氧化物酶(HRP)標記兔抗羊IgG和羊抗小鼠IgG由北京中杉公司進口分裝。聚偏二氟乙烯膜(PVDF)為 Milipore公司產品。溫度梯度PCR儀為德國Whatman Biometra公司產品。
1.2 方法
1.2.1 細胞培養與分組 常規方法復蘇腎小球系膜細胞后,采用胰酶消化法傳代、培養。將細胞分為3組:低糖組(LG組,給予5.5mmol/L葡萄糖刺激)、滲透壓對照組(LG+M組,給予5.5mmol/L葡萄糖+24.5mmol/L甘露醇)、高糖組(HG組,給予30mmol/L葡萄糖)。分別于刺激的24、48、72、96h收集細胞,提取蛋白及總RNA;同時收集細胞培養上清液。
1.2.2 Western blotting檢測 細胞用冰PBS(0.01mol/L)洗2遍,加入細胞裂解液冰浴1h,4℃、14 000r/min離心15min,取上清,Lowry法測定蛋白濃度。每上樣孔加75μg總蛋白行12%SDSPAGE凝膠電泳;濕轉法電轉移至PVDF膜,5%脫脂奶粉37℃封閉1h;加封閉液稀釋的一抗(1:1000)于4℃過夜;洗膜后加HRP標記的兔抗羊或羊抗小鼠IgG(1:5000)37℃孵育1.5h;洗膜后加化學發光劑ECL反應1min,曝光、沖洗后掃描蛋白條帶。以β-actin為內參照,采用美國UVP公司LabWork分析系統軟件對條帶進行定量分析。
1.2.3 RT-PCR檢測 采用Trizol試劑提取細胞總RNA,用紫外可見分光光度儀測定其純度和含量。2μg總RNA和oligo(dT)15引物按Promega公司反轉錄試劑盒要求加入試劑,反轉錄得到cDNA。取1μl cDNA進行PCR反應。所用引物、退火溫度、擴增片段長度及擴增條件如下。CTGF:正義5'-CTCGCCTTGGTGCTCCTCCT-3',反義5'-TGCGGTCCTTGGGCTCATC-3',擴增片段481bp,退火溫度60℃;MMP-2:正義5'-GGAGGCACGATTGGTCTG-3',反義5'-TTGGTT TCCGCATGGTCT-3',擴增片段299bp,退火溫度55℃;MT1-MMP:正義5'-CCTCAAGTGGCAGCATA AT-3',反義5'-AAGTTCTCGGTGTCCATCC-3',擴增片段459bp,退火溫度58℃;TIMP-2:正義5'-CCAAAGCAGTGAGCGAGAA-3',反義5'-CATCCAG AGGCACTCATCC-3',擴增片段386bp,退火溫度57℃;18S rRNA:正義5'-ACACGGACAGGATTGAC AGA-3',反義5'-GGACATCTAAGGGCATCACAG-3',擴增片段238bp,退火溫度56℃。所有引物均由上海生工生物公司合成,上述PCR反應均為30個循環。PCR產物行1.5%瓊脂糖凝膠電泳,以18S rRNA為內參照,在凝膠成像分析系統(美國UVP公司)下進行定量分析。
1.2.4 ELISA檢測 采用ELISA試劑盒檢測細胞培養上清中Ⅳ型膠原的含量,嚴格按照試劑盒說明書進行操作。
1.3 統計學處理 采用SPSS 12.0軟件進行統計學分析。計量資料符合正態分布,以表示,各組樣本方差齊,組間比較采用單因素方差分析,多重比較采用SNK-q檢驗,P<0.05為差異有統計學意義。
2 結 果
2.1 Western blotting檢測結果 與低糖組相比,高糖組MMP-2的表達在24h時略有升高,較低糖組增加10%±4%(P<0.05),至48h時則較低糖組減少42%±2%,其后隨時間延長表達持續降低,至96h時較低糖組減少78%±2%;MT1-MMP表達在24h開始下降并隨時間呈下降趨勢,高糖組與低糖組比較,在刺激的24h,MT1-MMP表達下降了29%±3%,隨后持續下降,至96h下降了78%±9%(P<0.01)。高糖組各時間點TIMP-2和CTGF表達均較低糖組上調,其中CTGF的表達在高糖刺激的24h即顯著上調,為低糖組的201%±24%,隨后持續增高,培養至96h為低糖組的484%±51%(P<0.01);TIMP-2的表達在24h時較低糖組增加55%±3%,且隨時間呈增高趨勢(P<0.01)。低糖組和滲透壓對照組組內、組間的各指標均無顯著性差異(圖1-4)。
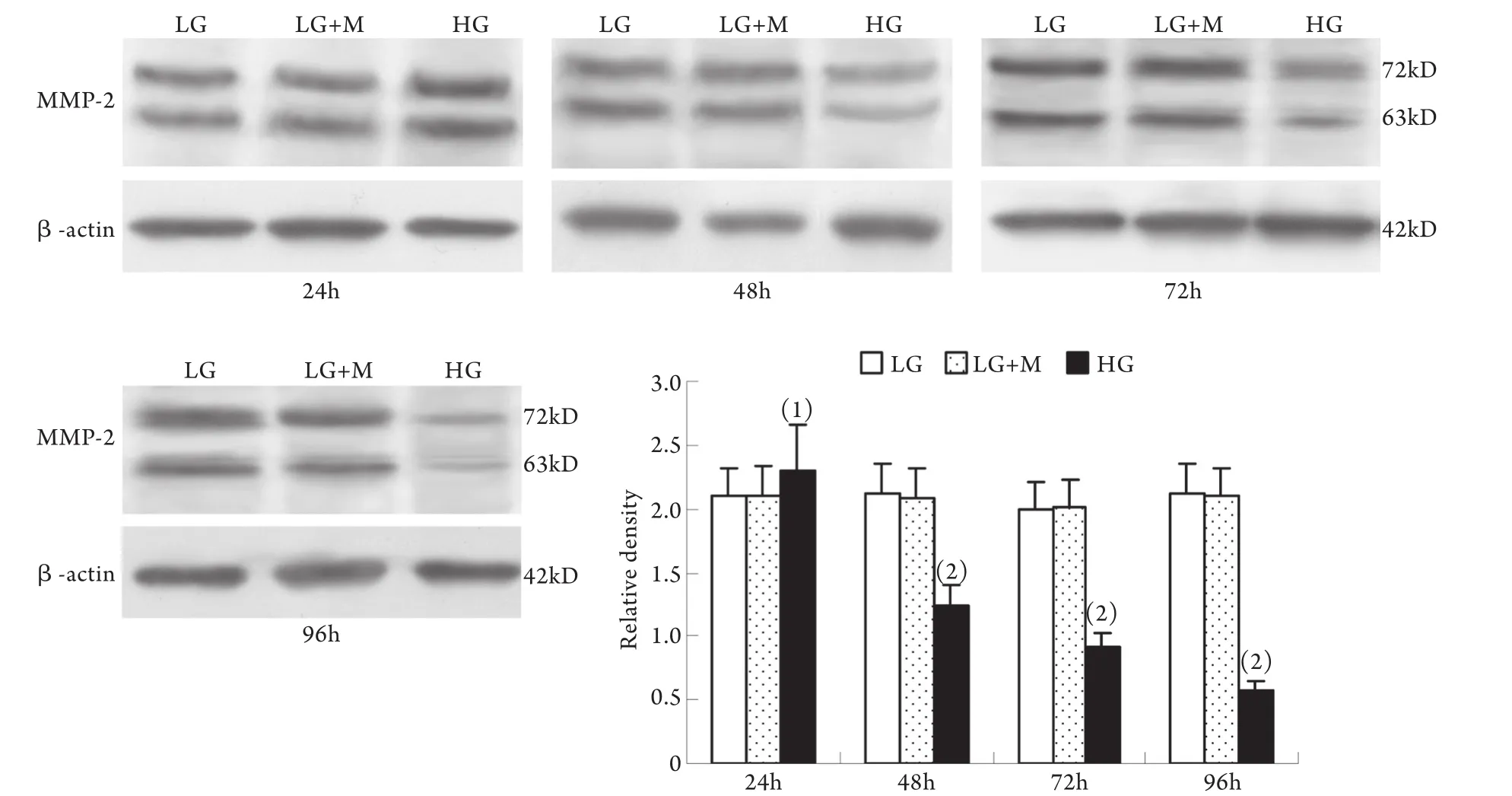
圖1 系膜細胞MMP-2蛋白表達結果Fig.1 Expression of MMP-2 protein in cultured GMCs
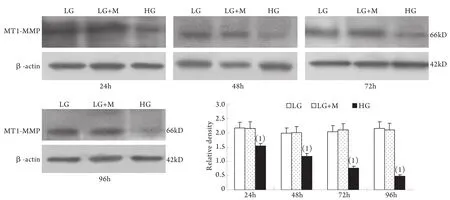
圖2 系膜細胞MT1-MMP蛋白表達結果Fig.2 Expression of MT1-MMP protein in cultured GMCs
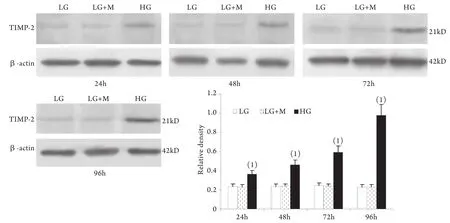
圖3 系膜細胞TIMP-2蛋白表達結果Fig.3 Expression of TIMP-2 protein in cultured GMCs
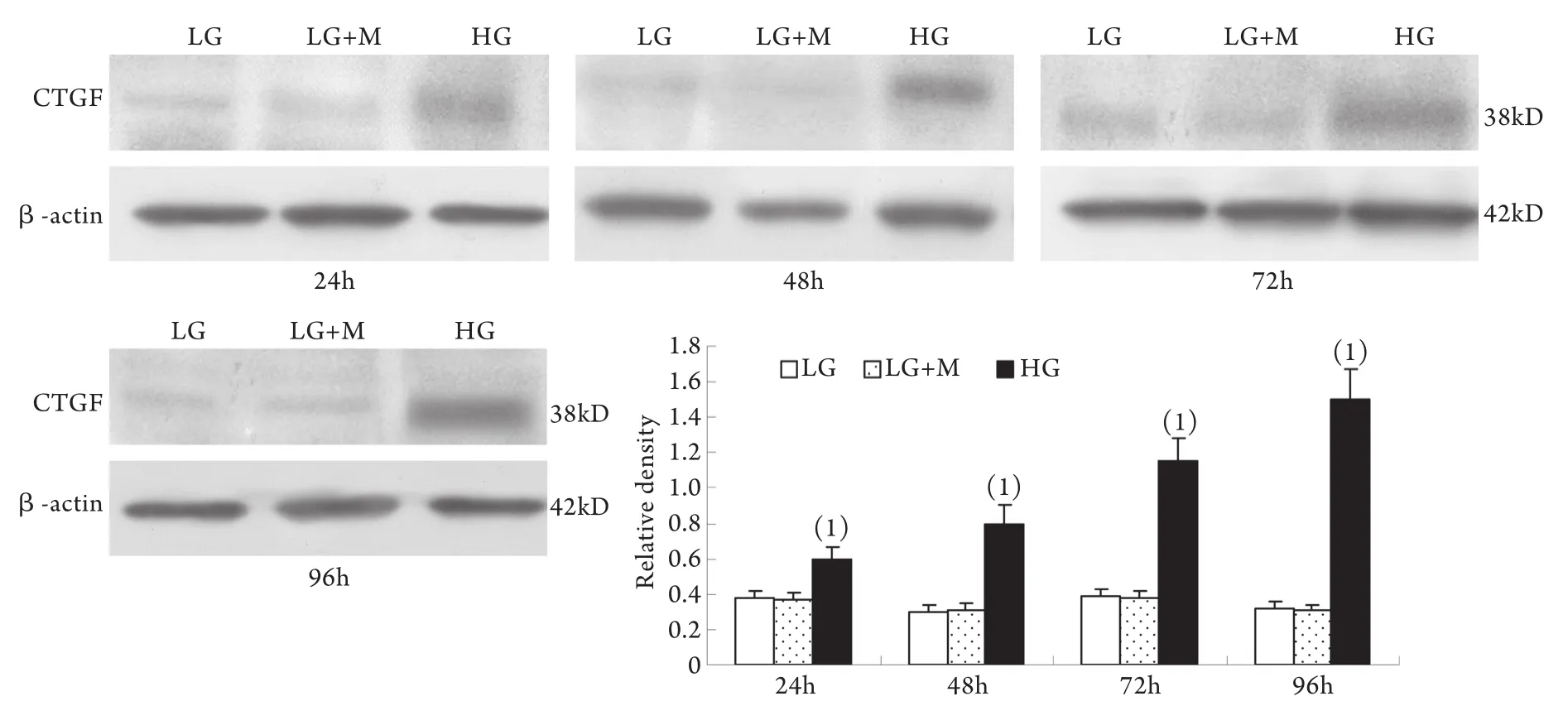
圖4 系膜細胞CTGF蛋白表達結果Fig.4 Expression of CTGF protein in cultured GMCs
2.2 半定量RT-PCR檢測mRNA的表達 MMP-2、TIMP-2、MT1-MMP和CTGF的mRNA表達與各自蛋白的表達趨勢基本一致。即CTGF mRNA和TIMP-2 mRNA在低糖組有少量表達,在高糖組各時間點表達上調,且隨時間呈增高趨勢,而MT1-MMP mRNA在低常糖組有較高表達,在高糖組各時間點表達下調且隨時間呈下降趨勢(P<0.01)。高糖組24h的MMP-2 mRNA表達略有增加,較低糖組增加9%±2%(P<0.05),其后48~96h則低于低糖組,且隨時間持續降低(圖5-8)。低糖組和滲透壓對照組組內、組間的各指標差異均無統計學意義。
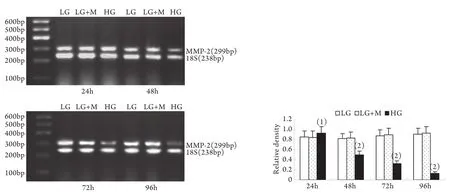
圖5 系膜細胞MMP-2 mRNA表達結果Fig.5 Expression of MMP-2 mRNA in cultured GMCs
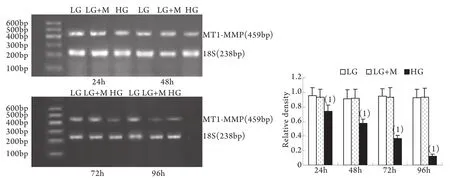
圖6 系膜細胞MT1-MMP mRNA表達結果Fig.6 Expression of MT1-MMP mRNA in cultured GMCs
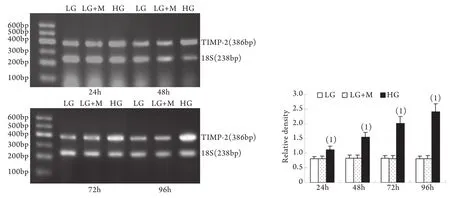
圖7 系膜細胞TIMP-2 mRNA表達結果Fig.7 Expression of TIMP-2 mRNA in cultured GMCs
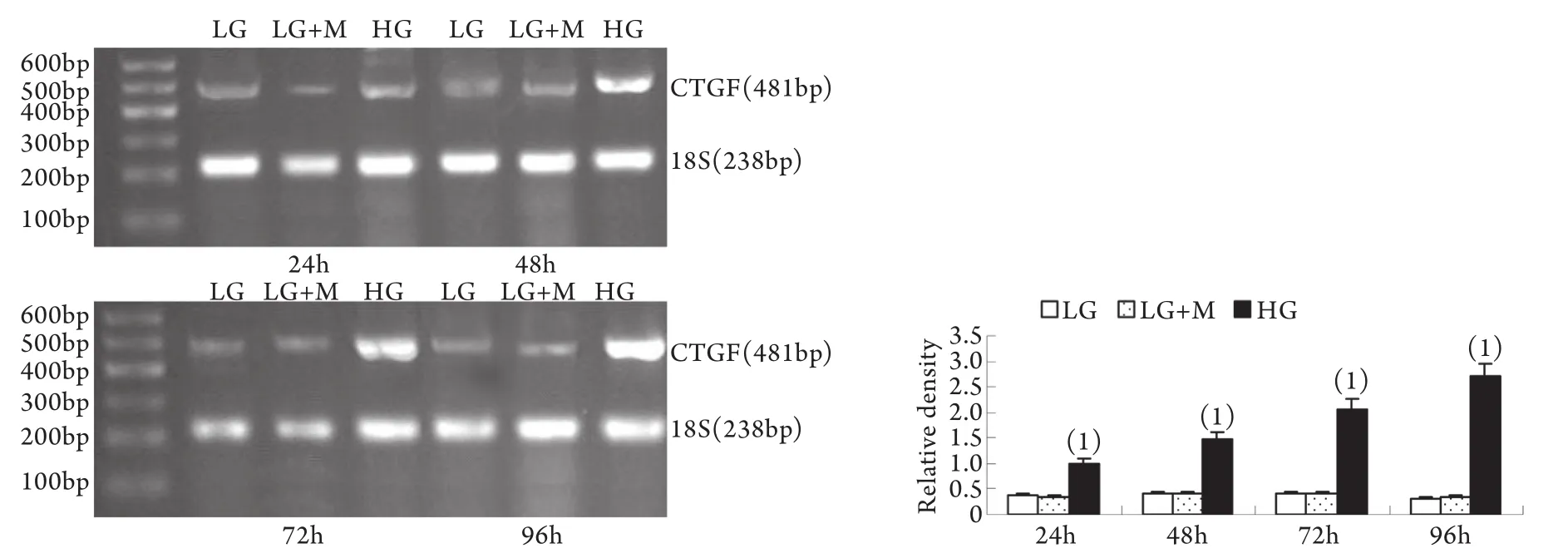
圖8 系膜細胞CTGF mRNA表達結果Fig.8 Expression of CTGF mRNA in cultured GMCs
2.3 細胞上清液檢測結果 與低糖組相比,高糖組細胞上清中的Ⅳ型膠原24h時即有增加,且持續增高至96h,低糖組和滲透壓對照組組內、組間的Ⅳ型膠原含量差異無統計學意義(P<0.05,表1)。
表1 不同時間點系膜細胞培養上清液中Ⅳ型膠原濃度(±s,μg/L)Tab.1 Concentration of collagen Ⅳ in the supernatants of GMCs at different time points±s, μg/L)
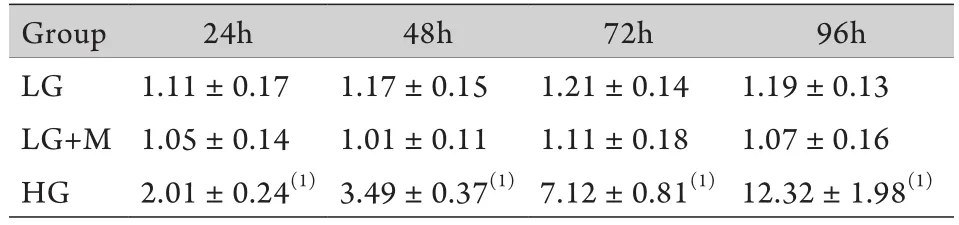
表1 不同時間點系膜細胞培養上清液中Ⅳ型膠原濃度(±s,μg/L)Tab.1 Concentration of collagen Ⅳ in the supernatants of GMCs at different time points±s, μg/L)
(1)P<0.01 compared with LG group and LG+M group
Group24h48h72h96h LG1.11±0.171.17±0.151.21±0.141.19±0.13 LG+M1.05±0.141.01±0.111.11±0.181.07±0.16 HG2.01±0.24(1)3.49±0.37(1)7.12±0.81(1)12.32±1.98(1)
3 討 論
腎小球系膜增生和擴張是DN的重要標志,并且系膜擴張程度與DN患者的腎功能和臨床癥狀密切相關。系膜擴張的主要原因是系膜基質合成、降解失衡。腎小球系膜細胞是腎小球中功能最活躍的細胞,具有合成細胞外基質、分泌細胞因子的功能,并能吞噬和清除異物,對于維持腎臟的正常組織結構和生理功能發揮著重要作用。早前一些體外研究顯示,高糖可誘導多種ECM成分的基因表達和蛋白合成[5-6]。近年來一些體內和體外實驗都提示DN中ECM降解過程受到抑制,且這種抑制現象與系膜細胞合成和分泌MMPs的功能異常有關[7-9]。多數體外實驗顯示高糖、血管緊張素Ⅱ(AngⅡ)、糖基化終產物(AGEs)和TGF-β1等刺激均可抑制系膜細胞MMP-9的表達和活性[10-13]。但是上述刺激因素對系膜細胞MMP-2及其相關抑制因子和激活因子的作用如何則報道不一,甚至相悖[9-15]。本實驗通過動態觀察高糖刺激對系膜細胞MMP-2的表達與活性的影響,發現高糖刺激對MMP-2的表達和活性的影響隨刺激時間的不同而變化。高糖刺激24h,系膜細胞MMP-2的基因表達、蛋白合成與酶活性均有小幅度增高,隨后從48h至96h MMP-2在轉錄、轉錄后翻譯和活化三個水平上均持續下降。與此同時系膜細胞分泌Ⅳ型膠原的量從24h開始增加,且持續增高至96h。這種在高糖刺激早期MMP-2的表達與活性上調現象的調節機制目前還不清楚,可能與高糖刺激系膜細胞ECM合成增多引起反饋性MMP-2表達上調有關[16],也可能與DN早期腎小球基底膜損傷和ECM重構有關[17]。這種系膜細胞MMP-2與Ⅳ型膠原變化大致反向平行的現象提示系膜基質降解減少可能是導致DN中腎小球系膜擴張和硬化的原因之一。
本實驗還同時觀察了高糖刺激對系膜細胞MT1-MMP、TIMP-2表達的影響,發現在低糖培養下腎小球系膜細胞有一定水平的MT1-MMP的表達,高糖刺激后,從24h開始MT1-MMP表達降低,并隨刺激時間延長持續降低。TIMP-2的變化則與MT1-MMP相反。從高糖刺激24h開始系膜細胞表達TIMP-2明顯升高,并隨刺激時間延長持續增高。MT1-MMP和TIMP-2 均參與MMP-2轉錄后的活性調節。MT1-MMP是MMP-2的特異膜型激活劑,主要表達于系膜細胞的胞膜部位,同時在胞質中也存在一定量可溶形式的MT1-MMP[18]。TIMP-2是MMP-2的特異組織抑制物,通過與MMP-2特異結合而阻止其對底物的降解作用。MT1-MMP和TIMP-2在系膜細胞的變化趨勢提示二者的異常變化可能是DN中MMP-2活性下降的原因之一。
目前研究普遍認為CTGF是TGF-β1最重要的下游效應因子[19],具有調節ECM代謝、促細胞增殖、遷移和誘導上皮-間質轉化的作用[20-21]。有體外研究顯示CTGF除具有促進ECM合成的作用外,還具有抑制基質成分降解的作用,這一作用部分通過抑制MMP-2的活化而實現[15]。本實驗在觀察高糖對系膜細胞MMP-2/TIMP-2和MT1-MMP的影響的同時,還檢測了系膜細胞中CTGF的表達變化。結果顯示高糖刺激使CTGF表達明顯增高,并且同系膜細胞分泌Ⅳ型膠原的變化趨勢相一致,與MMP-2的變化大致相反。此結果提示CTGF可能參與調節DN中ECM的降解過程。
總之,正常情況下系膜細胞、細胞因子、MMPs、MT1-MMP、TIMPs和ECM之間相互制約,使ECM 的合成和降解處于一個動態平衡,但當高糖存在時,這種正常的制約和平衡遭到破壞,最終導致ECM合成增加和(或)降解減少,引起系膜細胞ECM堆積,這是糖尿病腎病的病理基礎。
[1] Molitch ME, DeFronzo RA, Franz MJ,et al. Diabetic nephropathy[J]. Diabetes Care, 2003, 26 (Suppl 1): S94-S98.
[2] Thrailkill KM, Clay Bunn R, Fowlkes JL. Matrix metalloproteinases: their potential role in the pathogenesis of diabetic nephropathy[J]. Endocrine, 2009, 35(1): 1-10.
[3] Li M, Wang JS, Liu DW. Effect of mycophenolate mofetil on insulin-like growth factor-1 and matrix metalloproteinase-2 in the kidneys of diabetic rats[J]. J Zhengzhou Univ (Med Sci), 2013, 48(1): 77-80. [李敏, 王建生, 劉東偉. 霉酚酸酯對糖尿病大鼠腎組織中IGF-1、MMP-2 表達的影響[J]. 鄭州大學學報(醫學版), 2013, 48(1): 77-80.]
[4] Wang H, Zhao ZZ, Shen WQ,et al. Expressions of HIF-1α and CTGF in peritoneum of peritoneal dialysis rats[J]. J Zhengzhou Univ (Med Sci), 2012, 47(2): 205-208. [王紅, 趙占正, 申萬琴,等. 腹膜透析大鼠腹膜組織中HIF-1α和CTGF的表達[J]. 鄭州大學學報(醫學版), 2012, 47(2): 205-208.]
[5] Wang LH, Wu GL, Zhang LX,et al. p38 MAPK pathway in highglucose induced activation of rat mesangial cells[J]. Med J Chin PLA, 2009, 34(2): 196-199. [王麗暉, 吳廣禮, 張麗霞, 等. p38 MAPK信號途徑在高糖誘導的大鼠腎系膜細胞中激活的意義[J]. 解放軍醫學雜志, 2009, 34(2): 196-199.]
[6] Zhang D, Shao S, Shuai H,et al. SDF-1α reduces fibronectin expression in rat mesangial cells induced by TGF-β1 and high glucose through PI3K/Akt pathway[J]. Exp Cell Res, 2013, 319(12): 1796-1803.
[7] Takamiya Y, Fukami K, Yamagishi S,et al. Experimental diabetic nephropathy is accelerated in matrix metalloproteinase-2 knockout mice[J]. Nephrol Dial Transplant, 2013, 28(1): 55-62.
[8] Solini A, Rossi C, Santini E,et al. Angiotensin-II and rosuvastatin influence matrix remodeling in human mesangial cellsviametalloproteinase modulation[J]. J Hypertens, 2011, 29(10): 1930-1939.
[9] Yang XJ, Wang ZH, Li HC,et al. Correlation between matrix metalloproteinase-9 and Cystatin Cat the early stage of diabetic nephropathy[J]. Med J Chin PLA, 2006, 31(4): 346-348. [楊小潔, 王志慧, 李海潮, 等. 糖尿病腎病早期基質金屬蛋白酶-9與Cystatin C的相關性[J]. 解放軍醫學雜志, 2006, 31(4): 346-348.]
[10] McLennan SV, Kelly DJ, Cox AJ,et al. Decreased matrix degradation in diabetic nephropathy: effects of ACE inhibition on the expression and activities of matrix metalloproteinases[J]. Diabetologia, 2002, 45(2): 268-275.
[11] Dong FQ, Li H, Wu F,et al. Effects of overexpression of decorin on matrix metalloproteinases 2 and 9 in rat mesangial and tubular cells[J]. Zhonghua Yi Xue Za Zhi, 2008, 88(48): 3444-3447.
[12] Ohtomo S, Nangaku M, Izuhara Y,et al. The role of megsin, a serine protease inhibitor, in diabetic mesangial matrix accumulation[J]. Kidney Int, 2008, 74(6):768-774.
[13] Sun SZ, Wang Y, Li Q,et al. Effects of benazepril on renal function and kidney expression of matrix metalloproteinase-2 and tissue inhibitor of metalloproteinase-2 in diabetic rats[J]. Chin Med J (Engl), 2006, 19(10): 814-821.
[14] Lee MP, Sweeney G. Insulin increases gelatinase activity in rat glomerular mesangial cellsviaERK- and PI-3 kinase-dependent signaling[J]. Diabetes Obes Metab, 2006, 8(3): 281-288.
[15] McLennan SV, Wang XY, Moreno V,et al. Connective tissue growth factor mediates high glucose effects on matrix degradation through tissue inhibitor of matrix metalloproteinase type 1: implications for diabetic nephropathy[J]. Endocrinol, 2004, 145(12): 5646-5655.
[16] Corcoran ML, Hewitt RE, Kleiner DE,et al. MMP-2: expression, activation and inhibition[J]. Enzyme Protein, 1996, 49(1-3): 7-19.
[17] Keeling J, Herrera GA. Matrix metalloproteinases and mesangial remodeling in light chain-related glomerular damage[J]. Kidney Int, 2005, 68(4): 1590-1603.
[18] Yao M, Zeng S, Tu Y,et al. Changes of expression and activity of renal matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases in the stroke-prone spontaneously hypertensive rats[J]. J Logist Univ CAPF (Med Sci), 2013, 22(3): 161-165. [姚旻, 曾山, 涂悅, 等. 高血壓大鼠腎臟基質金屬蛋白酶及組織型基質金屬蛋白酶抑制劑表達與活性的變化[J]. 武警后勤學院學報(醫學版), 2013, 22(3): 161-165.]
[19] Boor P, Floege J. Chronic kidney disease growth factors in renal fibrosis[J]. Clin Exp Pharmacol Physiol, 2011, 38(7): 391-400.
[20] Russo JW, Castellot JJ. CCN5: biology and pathophysiology[J]. J Cell Commun Signal, 2010, 4(3): 119-130.
[21] Mason RM. Connective tissue growth factor (CCN2), a pathogenic factor in diabetic nephropathy. What does it do? How does it do it[J]? J Cell Commun Signal, 2009, 3(2): 95-104.
Expression of MMP-2, TIMP-2, CTGF and MT1-MMP in rat glomerular mesangial cells stimulated by high-glucose and its significance
YAO Fang1, LI Zhi-hong2, YAN Zhe1, LIU Qing-juan1, DUAN Hui-jun1, QIE Tao21Department of Pathology, Hebei Medical University, Shijiazhuang 050017, China
2Department of Endocrinology, First Central Hospital of Baoding City, Baoding, Hebei 071000, China
This work was supported by the Youth Foundation of Education Department of Hebei Province (2010150)
ObjectiveTo observe the dynamic changes of matrix metalloproteinase-2 (MMP-2), tissue inhibitor of metalloproteinase-2 (TIMP-2), membrane-type 1 matrix metalloproteinase (MT1-MMP) and connective tissue growth factor (CTGF) expression in high glucose-stimulated glomerular mesangial cells (GMCs) in rats, and investigate the mechanism of the pathogenesis of diabetic nephropathy.MethodsRat HBZY-1 GMCs were cultured and divided into 3 groups: low concentration (5.5mmol/L) D-glucose (LG) group, high concentration (30mmol/L) D-glucose (HG) group and 24.5mmol/L mannitol plus 5.5mmol/L D-glucose (LG+M) group (served as osmotic pressure control). The mRNA and protein expressions of MMP-2, TIMP-2, MT1-MMP and CTGF were detected with semi-quantitative RT-PCR and Western blotting, and the secreted collagen Ⅳin supernatants of the GMCs was detected by ELISA after cultured for 24, 48, 72 and 96h.ResultsCompared with LG group, after exposure to high glucose for 24h, MMP-2 expression was slightly increased (increased by 10%±4%) in GMCs in HG group (P<0.05). But when the exposure time last from 48h to 96h, the expression of MMP-2 was decreased by 42%±2% to 78%±2%(P<0.01). Compared with LG group, high-glucose incubation resulted in down-regulation of MT1-MMP (decreased by 29%±3% at 24h to 78%±9% at 96h,P<0.01) whereas up-regulation of TIMP-2 (increased by 55%±3% at 24h,P<0.01) and CTGF (increased by 201%±24% at 24h to 484%±51% at 96h,P<0.01). RT-PCR revealed consistent dynamic changes of MMP-2, TIMP-2, MT1-MMP and CTGF with their protein changes listed above. Compared with LG group, the secreted collagen Ⅳ in supernatants of HG group was increased by 201%±24% at 24h to 1232%±198% at 96h (P<0.01). There was no significant difference of above indexes between LG group and LG+M group.ConclusionsHigh glucose may induce and activate MMP-2 transiently, but can inhibit the expression of MMP-2 and MT1-MMP for a long term whereas induce the expressions of TIMP-2 and CTGF in GMCs. MMP-2, TIMP-2, MT1-MMP and CTGF may contribute to the metabolism imbalance of extracellular matrix then induce the accumulation of extracellular matrix in diabetic nephropathy.
diabetic nephropathy; glomerular mesangial cells; metalloproteinase-2; tissue inhibitor of metalloproteinase-2; connective tissue growth factor; membrane-type 1 matrix metalloproteinase
R587.1
A
0577-7402(2013)10-0811-07
10.11855/j.issn.0577-7402.2013.10.006
2013-05-14;
2013-07-15)
(責任編輯:張小利)
河北省教育廳青年基金(2010150)
姚芳,醫學博士。主要從事糖尿病腎病基礎醫學研究
050017 石家莊 河北醫科大學基礎醫學院病理教研室(姚芳、閆喆、劉青娟、段惠軍);071000 河北保定 保定市第一中心醫院內分泌科(李志紅、郄濤)

