Effectiveness and safety of generic memantine hydrochloride manufactured in China in the treatment of moderate to severe Alzheimer’s disease: a multicenter, double-blind randomized controlled trial
Minjie ZHU, Shifu XIAO*, Guanjun LI, Xia LI, Mouni TANG, Siming YANG, Xiufeng XU,Lianyuan FENG, Kaixiang LIU, Lianping HU
?Original article?
Effectiveness and safety of generic memantine hydrochloride manufactured in China in the treatment of moderate to severe Alzheimer’s disease: a multicenter, double-blind randomized controlled trial
Minjie ZHU1, Shifu XIAO1*, Guanjun LI1, Xia LI1, Mouni TANG2, Siming YANG3, Xiufeng XU4,Lianyuan FENG5, Kaixiang LIU6, Lianping HU7
1. Introduction
Alzheimer’s disease (AD) is a progressive degenerative disease of the central nervous system with marked memory impairment and agnosia, declines in daily functioning, and associated psychiatric and behavioral symptoms. The development of AD is a complex process involving the toxic effects of some excitatory amino acids, particularly glutamate – the most common excitatory neurotransmitter in the human brain which is closely related to cell death in the brains of individuals with dementia.[1]This toxicity is mainly mediated by N-methyl-D-aspartate (NMDA) glutamate receptors (a subtype of glutamate receptors) that play an important role in the early development of the nervous system, in the transmission of excitatory neurotransmitters within the central nervous system, in the plasticity of neuronal synapses, and in memory and learning.[2]
Currently, there is no effective cure for AD so the focus of treatment is on stopping or slowing the progressive decline in cognitive functioning.[3]Memantine is a noncompetitive NMDA receptor antagonist which may slow down the neurodegenerative process of AD.Memantine blocks hyperactive NMDA receptors and,thus, can protect nerve cells by inhibiting Aβ deposition,decreasing the phosphorylation of tau proteins, and reducing abnormal synaptic signals and neuronal cell damage.[4]A 28-week multicenter, double-blind randomized placebo-controlled study conducted in the United States found that the progressive deterioration of moderate to severe AD could be alleviated with a daily oral dose of 20 mg memantine hydrochloride.[5]A subsequent 24-week open-label extension study showed that all of the measures of efficacy considered in the study improved significantly in the individuals who had received placebos when they were subsequently treated with memantine hydrochloride.[6]Based on these findings,in 2003 memantine hydrochloride was approved by the United States Food and Drug Administration for the treatment of moderate to severe AD.[7,8]Other studies have reported that it is also effective for mild AD.[9,10]
Clinical trials suggest that continuous treatment with menantine can improve cognitive and daily functioning in patients with AD,[11]but the long-term use of this medication can be quite costly. This can be a major financial burden for patients and their families or for national health insurance systems in countries with rapidly aging populations. To reduce the cost of treating this increasingly prevalent condition in China, the State Food and Drug Administration of China approved clinical trials of a domestically manufactured generic version of memantine hydrochloride (jointly developed by Anhui Huachen Pharmaceutical Ltd and Bio-technology Ltd of the University of Science and Technology of China;approval number 2005L02694). If this generic form of the medication is approved it will cost less than half that of the imported proprietary form.
The current randomized controlled trial was conducted in six centers in China from 1 February 2009 to 28 July 2010 to assess the efficacy and safety of domestically manufactured (i.e., generic) memantine hydrochloride tablets in the treatment of moderate to severe Alzheimer’s disease.
2. Methods
2.1 Sample
The six participating centers in the study included two specialty psychiatric hospitals, the psychiatric department of a general hospital and three neurology departments in general hospitals. The enrolment of subjects for the study is shown in Figure 1. Inclusion and exclusion criteria were as follows:
? outpatient at one of the participating centers;
? 45 to 85 years of age;
? meets diagnostic criteria for AD of the American Psychiatric Association Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSMIV);[12]
? has moderate to severe AD based on the Mini-Mental State Examination (MMSE)[13](i.e., a total score >5 and a score of <20 for patients with an education above primary school, <17 for those who only completed primary school, and <14 for those who did not complete primary school);
? does not have ischemic dementia (based on clinical and laboratory tests, brain imaging study completed within the prior year, and a Hachinski Ischemic Scale[14]score <4);
? does not have physical disorders (e.g., intracranial lesions) endocrine diseases, (e.g., hypothyroidism)or substance abuse problems that could cause the dementia;
? does not have serious depressive symptoms(Hamilton Depression Scale[15]score < 10);
? does not have a serious physical illness;
? does not use cognitive-boosting medications(including co-dergocrine mesyiate tablets, Duxil,Acetamide Pyrrolidone, aniracetam, etc.) within two weeks of entering the trial;
? did not participate in another AD treatment trial within the prior 3 months;
? no known allergy to memantine hydrochloride;
? difference in screening and baseline MMSE score is less than three points;
? and patient’s legal guardian provided written informed consent
2.2 Method
This trial, which was approved by China’s FDA and the Ethics Committee of the Shanghai Mental Health Center, was conducted in six centers in China. The Hefei Zhongkeda Biological and Technology Company provided both the trial drug (domestic memantine hydrochloride tablets, 10 mg/ tablet, batch number 0810001), andthe imported proprietary form of the drug (memantine hydrochloride tablets, Ebixa, 10 mg/tablet, batch number 7088461). The generic and proprietary forms of the medication were the same in color, shape, fl avor,and size. Enrolled patients at each of the six centers were randomly assigned to the generic medication group or the proprietary medication group using a computergenerated random number list. The generic medication group took half a pill per day for the first week, 1 pill per day for the second week, 1.5 pills per day for the third week, and 2 pills per day from the 4thto the 16thweek.The control group took the imported proprietary form of memantine hydrochloride tablets at the same dosage.All the participants were outpatients; the medications were administered by co-resident family members.
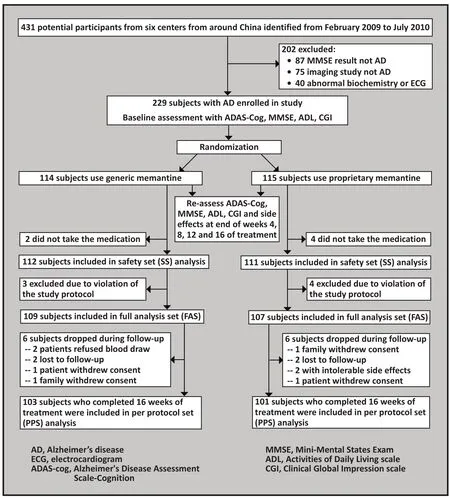
Figure 1. Flowchart of the study
2.3 Assessment
2.3.1 Assessment of treatment effect
The primary outcome was the change in scores of the cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-Cog).[16]The secondary outcomes were changes in scores on the MMSE, Activities of Daily Living(ADL) scale,[17]and Clinical Global Impression (CGI)[18]scale. All four scales were rated at baseline and at the end of the 4th, 8th, 12th, and 16thweek after initiating treatment; the ratings were conducted by a psychiatrist or a neurologist who had been trained in the use of the scales and who were blind to the treatment group of the subjects.
ADAS-Cog assesses 12 different aspects of cognitive functioning (including memory, attention, language,orientation, visuospatial skills, understanding, and operationalization); the range in scores are from 0 to 75 with lower scores representing better functioning. Using a score of 15.5 as the cut-off score, the Chinese version of the ADAS-Cog can discriminate individuals with AD from normal controls (sensitivity=92%, specificity=90%,area under ROC=0.95).[19,20]The MMSE is a 30-item scale that assesses orientation to time and place, immediate and short-term memory, language, and other cognitive functions; the total score ranges from 0 to 30 with higher scores representing better functioning. The 14-item ADL is used to assess daily functioning; the total score ranges from 14 to 56 with lower scores representing better functioning. The CGI has three items on 0 to 7 Likert scales (higher scores represent better functioning) that assess current disease severity, total effect of treatment,and an effect index adjusted for the severity of sideeffects.
2.3.2 Safety indices
Vital signs and adverse events were recorded at each visit.Routine blood tests, urine tests, EKG, blood biochemistry(liver and kidney function, electrolytes, glucose, etc.)tests were conducted at the end of the 8thand 16thweeks of treatment.
2.4 Statistical analysis
SAS 9.2 statistical software was used for statistical analysis. The data at baseline were analyzed using a safety set (SS) analysis that included all enrolled subjects who used any medication (excluding 6 randomized patients who did not start medication). The main measurement outcomes were assessed using the full analysis set (FAS) that included all subjects who followed the protocol; this analysis was used to determine the final conclusion of the trial. For participants who started treatment but did not complete the entire treatment,the Last Observation Carried Forward (LOCF) method was applied in the FAS analysis, which ensured that the number of cases at each time point was the same.A per protocol set (PPS) analysis limited to subjects who completed the 16-week intervention according to the protocol was also conducted. Two-sample t-tests, rank sum tests and paired t-tests were used to compare the main outcomes between groups at four follow-up points, and to estimate differences from the baseline. Adverse events were tabulated and the total incidence was comparison between the two groups using chi-square test or Fisher’s exact test. All statistical tests were two-sided, and results were considered statistically significant when the p-value was <0.05.
3. Results
A total of 431 potential subjects were identified from the six sites (from 60 to 87 individuals from each site)and 229 of them who met the enrollment criteria were randomized (using a random number sequence generated by a software package) within each center either to the generic medication group (n=114) or to the proprietary medication group (n=115). After blinded review of screening data, 13 randomized subjects were excluded due to violation of the study protocol: 5 subjects were excluded from the generic medication group either because their MMSE scores changed more than 2 points between screening and baseline (n=3) or because they did not start taking the study medication (n=2); 8 patients were excluded from the proprietary medication group either because the MMSE score changed more than 2 points between screening and baseline (n=1),because the screening MMSE score did not meet enrolment criteria (n=3), or because the patient did not start taking the study medication (n=4). Thus this left 109 patients in the generic medication group and 107 in the proprietary medication group at the beginning of the treatment. As shown in Figure 1, 6 individuals dropped out of each group over the 16 weeks of the trial, so 103 generic medication group subjects and 101 proprietary medication group subjects completed the full course of treatment. Thus, 223 subjects were included in the SS analysis, 216 in the FAS analysis and 204 in the PPS analysis.
3.1 Comparison of the baseline characteristics of the two groups
As shown in Table 1, the SS analysis found no statistically significant differences between the two groups at baseline in gender, age, educational level, baseline vital signs, Hachinski Ischemic Index, HAMD-17 score, or MMSE score. There were also no statistically significant differences between the groups in the mean baseline ADAS-Cog score and mean ADL score among subjects who followed the study protocol (using the FAS analysis).Repetition of this analysis of baseline values in patients who completed the 16 weeks of treatment (the PPS analysis) also found no statistically significant differences between the groups
3.2 Comparison of efficacy
3.2.1 Primary outcome
As shown in Tables 2 and 3 and Figure 2, between-group differences in ADAS-Cog scores were not statistically significant at any of the time points considered in the study. For both groups the ADAS-Cog score decreased significantly from the baseline score at the end of the 4th,8th, 12thand 16thweeks of treatment; the magnitude of the drop in the score at these four time periods was not significantly different between the two groups. A parallel analysis using the PPS had the same results.
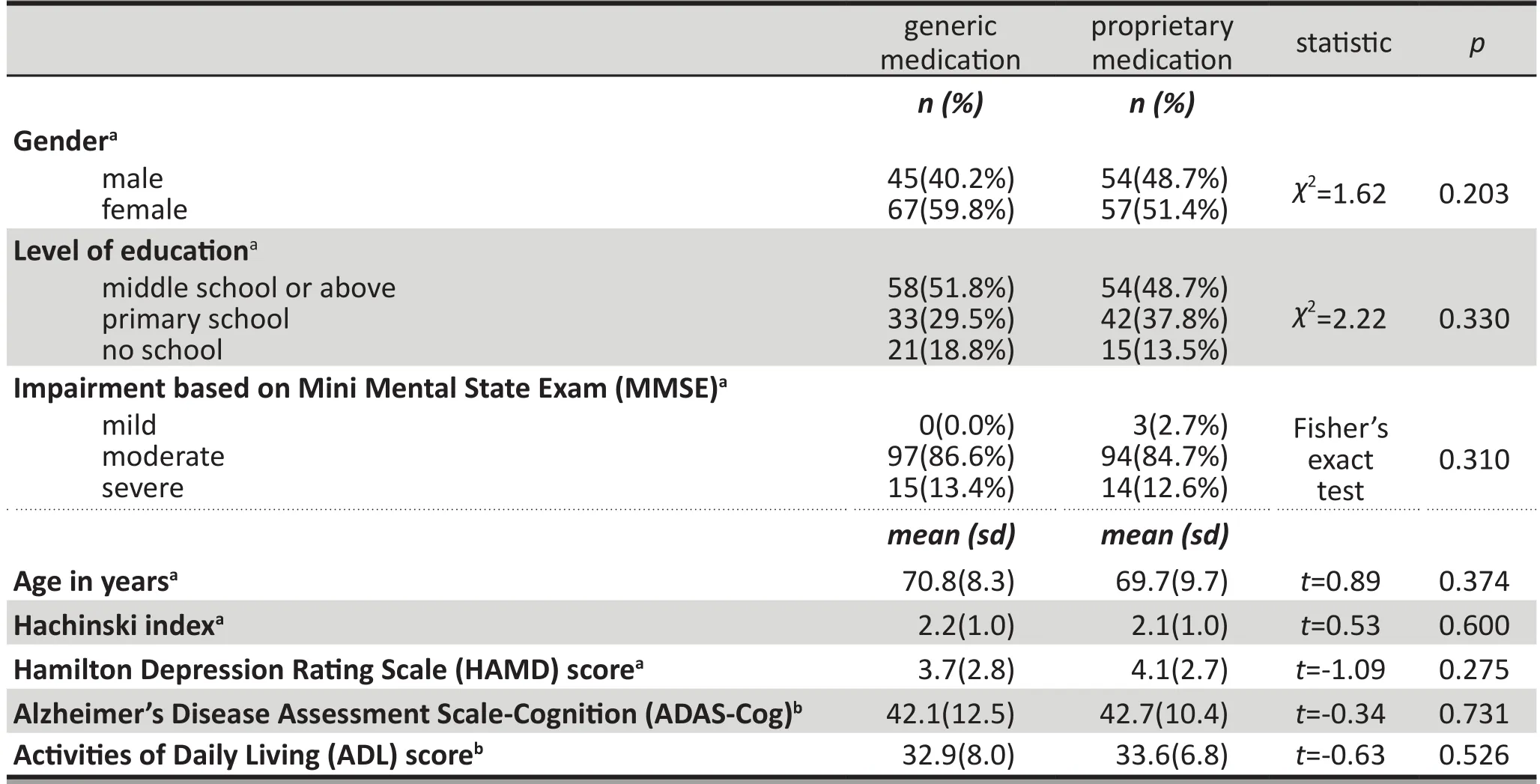
Table 1. Baseline characteristics of participants enrolled in the study
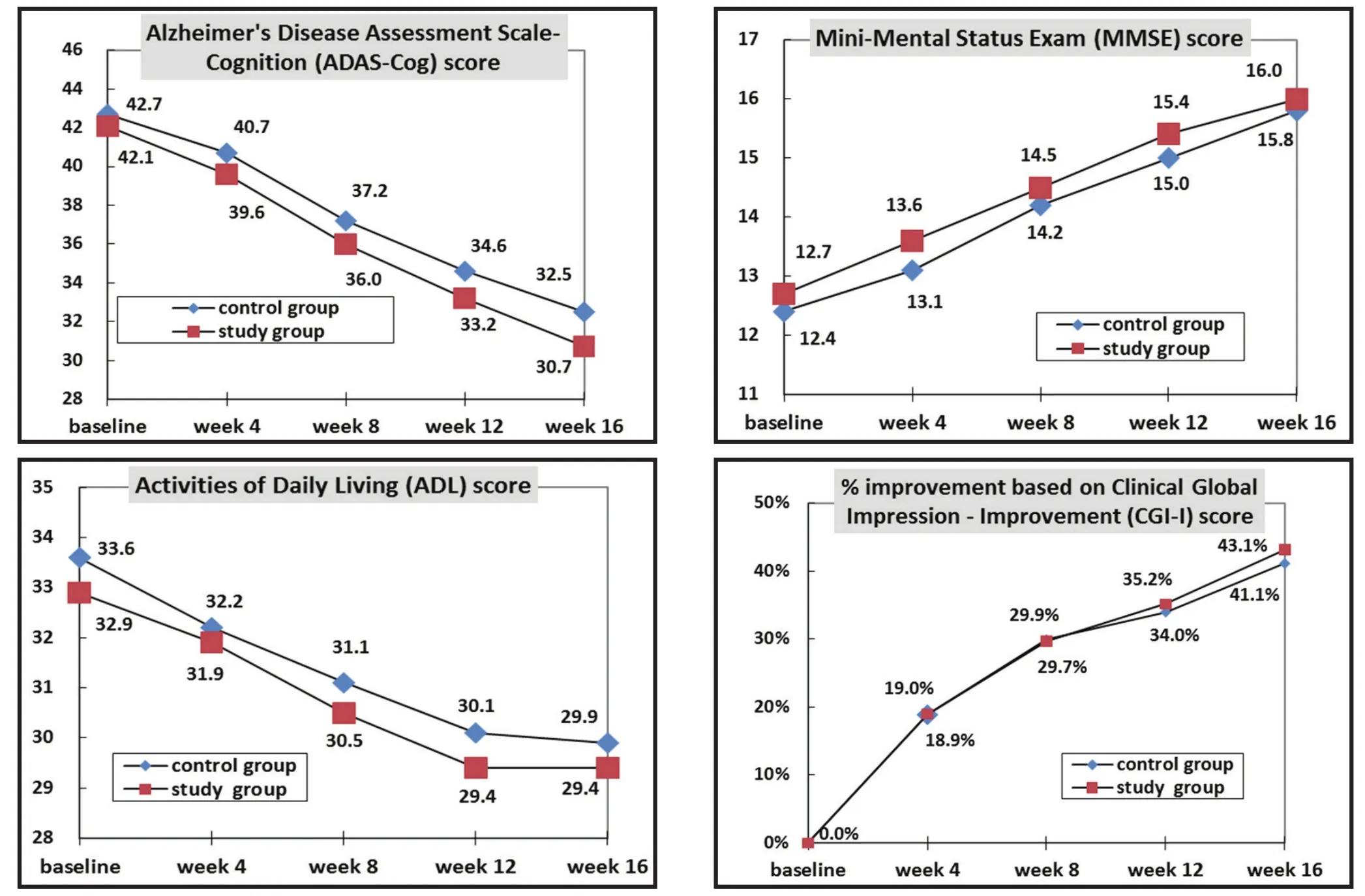
Figure 2. Comparison of outcome measures over the 16-week trial in individuals with Alzheimer’s Disease who take generic memantine (study group) or proprietary memantine (control group)
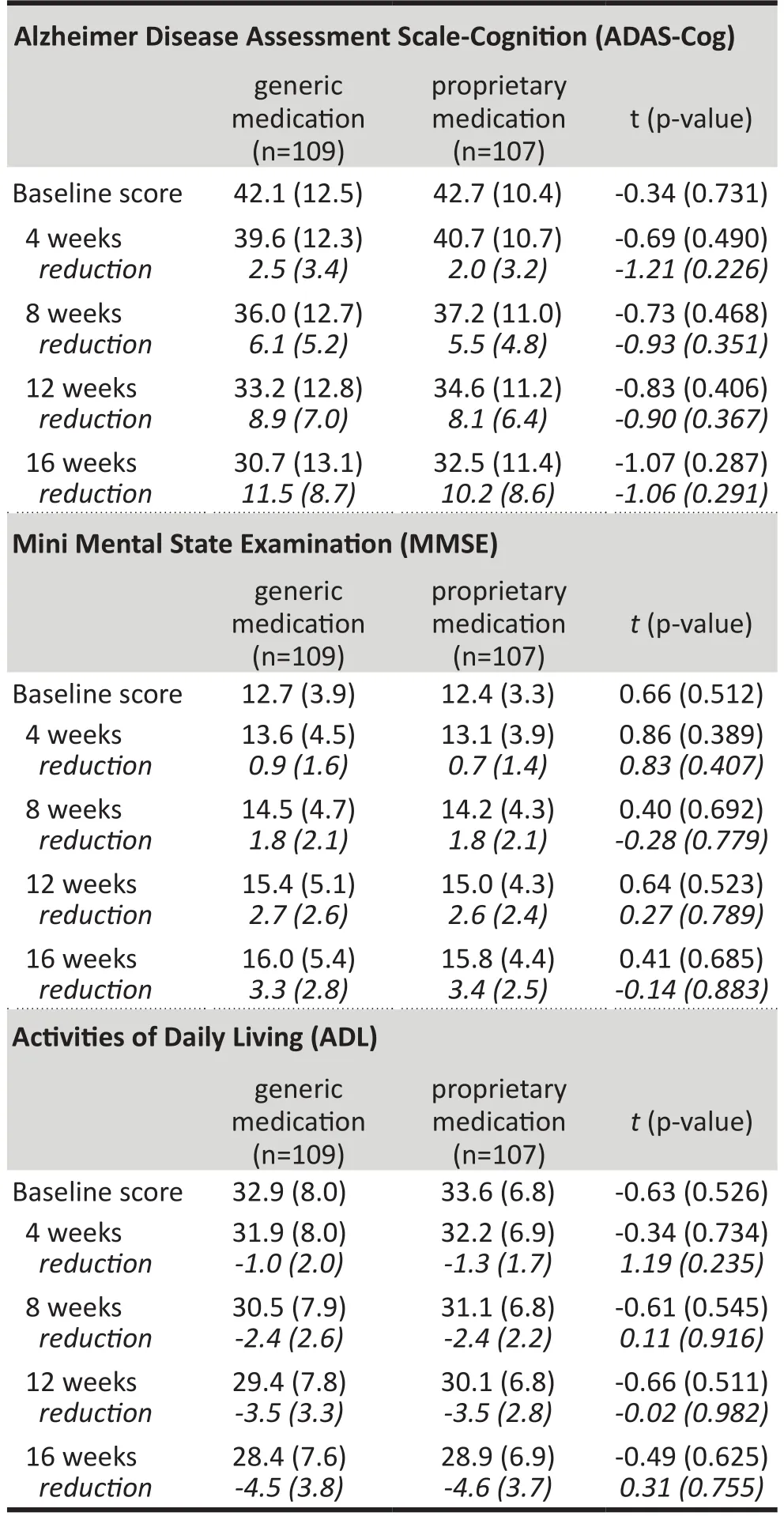
Table 2. Comparison of mean (sd) scores on cognitive tests between patients taking generic and proprietary memantine at each follow-up evaluation using the Full Analysis Set (FAS)
3.2.2 Secondary outcomes
Tables 2 and 3 and Figure 2 also show the results for the MMSE and ADL scales. There were not statistically significant differences between the generic medicationgroup and the proprietary medication group at any of the time periods assessed. The MMSE increased significantly over time in both groups and the ADL decreased significantly over time in both groups. There were no significant differences in the magnitude of these changes over time between the two groups. Analysis using the PPS produced the same results.
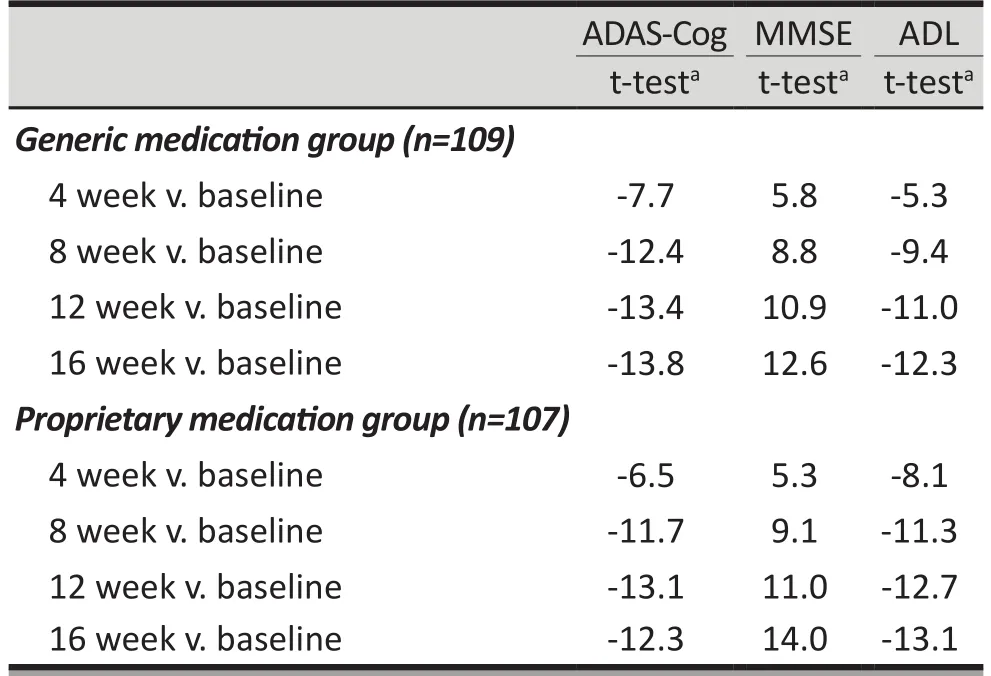
Table 3. Within-group changes over time of scores on cognitive tests for patients with Alzheimer’s disease taking generic and proprietary memantine using the Full Analysis Set (FAS)
The changes in CGI scores over the course of treatment are shown in Figure 2. Based on the FAS analysis of the CGI scores, at the end of the 16 weeks of treatment the proportions of subjects in the generic medication and proprietary medication groups who were ‘significantly improved’ (0.0% and 0.9%,respectively), ‘moderately improved’ (40.4% and 32.8%),‘mildly improved’ (48.6% and 55.1%), and unchanged or worsened (11.0% and 11.2%) were not statistically different between the two groups.
3.3 Safety analysis
Two cases of serious adverse events occurred during the trial. One subject in the generic medication group died suddenly after randomization but before starting the study medication. One subject in the proprietary medication group was hospitalized due to multiple cerebral infarctions during the follow-up period; he was subsequently dropped from the study. Neither of these events was directly or indirectly related to use of the trial medication.
The most common adverse events are shown in Table 4. Based on the SS analysis, there were 23 subjects in the generic medication group (20.5%) who experienced adverse events and 30 subjects in theproprietary medication group (27.0%) who experienced adverse events (χ2=1.30, p=0.255). Most of these adverse events were probably not directly related to medication usage but, rather, the common symptoms seen in elderly individuals.
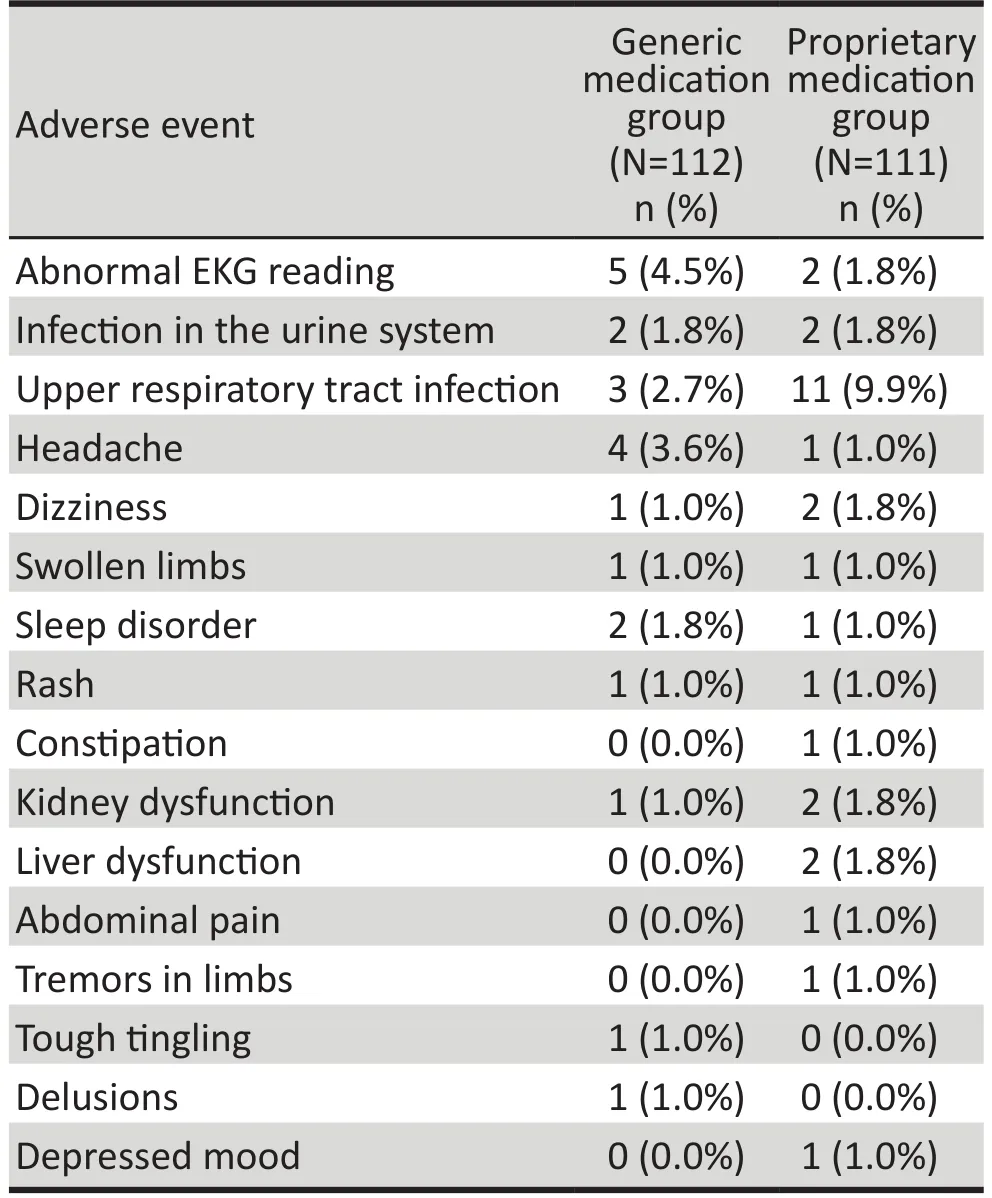
Table 4. Occurrence of adverse events in patients with Alzheimer’s disease treated with generic and proprietary memantine based on the Safety Set(SS) analysis
Seven individuals in the generic medication group(6.3%) and nine individuals in the proprietary medication group (8.1%) experienced adverse events that were considered related to the medication (χ2=0.29, p=0.591).In the generic medication group, three individuals had EKG abnormalities and one each had bipedal edema, eczema, insomnia, and renal dysfunction. In the proprietary medication group two individuals had abnormal liver function tests, two had renal dysfunction,and one each had sinus bradycardia, constipation,abdominal pain, rash, and trembling limbs. In most cases these adverse reactions were mild and resolved on their own without any specific treatment. The severity of adverse events was mostly mild and did not require special treatment. There were no statistically significant differences in vital signs, laboratory tests or EKG results between the two groups at any point in time throughout the study.
4. Discussion
4.1 Main findings
This multi-center study enrolled a large sample of individuals with AD from different parts of China and used randomized, double-blind methods to compare the efficacy and safety of domestically produced generic memantine to that of imported proprietary memantine.Changes in ADAS-Cog total scores among individuals with moderate to severe AD before treatment versus 16 weeks after treatment were used as the primary indicators for the treatment effect. Based on this measure, both forms of the medication resulted in significant improvement in the cognitive functioning of study participants. Moreover,there were no significant differences in the magnitude of improvement or in the rate of improvement between the two forms of the medication. Results for the three secondary measures of outcome assessed – changes in MMSE scores, ADL scores, and CGI scores – were the same. The efficacy of memantine found in this study confirms the results of many studies in other countries.[11,21,22]The current study also shows that the generic form of the medication manufactured in China is equally effective as the imported proprietary form of the medication.
In the current study two participants (one receiving generic medication and one receiving proprietary medication) experienced serious adverse events (one sudden death and one multi-infarct stroke), but it appears doubtful that these adverse events were related to use of memantine. The adverse events that were clearly related to the medication (including abnormal EKG readings, bipedal edema, eczema, insomnia, and kidney dysfunction) were mild and remitted spontaneously.There was, moreover, no significant difference in the prevalence or severity of these adverse reactions between the generic medication and proprietary medication groups. Thus these results support the findings of previous studies that found memantine hydrochloride to be reasonably safe and well tolerated in AD patients.[6,22-24]
4.2 Limitations
There are several potential limitations to these results.As shown in Table 2 and in Figure 2, there was a clear stepwise improvement in the four outcome measures at each of the four times the individuals were followed up during the 16-week study; we analyzed results at each time point and found no difference between the two groups but we did not conduct a repeated measures analysis of variance analysis to demonstrate that the overall trend in changes during the 16 weeks did not vary between the generic medication group and the proprietary medication group. Patients with AD may be advised to take memantine indefinitely, so a 16-week trial may not be long enough to identify the long-term efficacy or late-appearing adverse reactions of the medication,though it’s doubtful that there would be substantial differences in the long-term efficacy and safety between the generic and proprietary forms of the medication.One important consideration not addressed in the current study is the relative cost of the two forms of the medication; cost-benefit analyses need to be included in future longitudinal studies that compare the generic and proprietary forms of memantine. Another area that has not been explored in detail is the appropriate dosing and intervals between doses; further study may help to individualize the dosage and frequency of administration of memantine based on patient characteristics.
4.3 Significance
This study convincingly demonstrates that the generic form of memantide produced in China is equivalent to the imported proprietary form of memantine with respect to efficacy, tolerability, and safety over the first 16 weeks of treatment. Further study is needed to confirm the efficacy and safety of the long-term use of the generic form of the medication. Given the substantially lower price of the generic form of memantine and the expectation that individuals with AD should take this medication continuously, the widespread use of generic forms of the medication could substantially reduce the financial burden of families who are caring for elders with AD and decrease the health care costs of governments in jurisdictions that have rapidly aging populations.
Funding
The study was jointly funded by Major New Medication Development Program (2012ZX09303005-002) and the National Key Clinical Disciplines at Shanghai Mental Health Center (Office of Medical Affairs, Ministry of Health, 2011-873; OMA-MH, 2011-873). The medications used in the study were provided by the Hefei Zhongkeda Biological and Technology Company. The use of these medications was approved by the Chinese Food and Drug Administration (2005l02694).
Conflict of interest
The authors report not financial or other conflict of interest related to the study.
Acknowledgment
The authors would like to sincerely thank the patients who participated in the study and their care-givers.We would also like to thank the clinicians, researchers and interviewers at each of the participating centers who conducted the evaluation and treatment of study participants: the Shanghai Mental Health Center,Guangzhou Psychiatric Hospital, First People’s Hospital of Chenzhou, First Affiliated Hospital of Kunming Medical School, Chinese People’s Liberation Army Bethune International Peace Hospital, and the Affiliated Hospital of Guilin Medical School. We also thank the Venturepharma Center of Data Management and Analysis for their assistance in the analysis of the results.
1. Wenk GL. Neuropathologic changes in Alzheimer’s disease:potential targets for treatment. J Clin Psychiatry 2006;67(suppl3): 3-6.
2. Johnson JW, Kotermanski SE. Mechanism of action of memantine. Curr Opin Pharmacol 2006; 6: 61-67.
3. Wilkinson D, Andersen HF. Analysis of the effect of memantine in reducing the worsening of clinical symptoms in patients with moderate to severe Alzheimer’s disease.Dement Geriatr Cogn Disord 2007; 24(2): 138-145
4. Gunnarsson MD, Kilander L, Basun H, Lannfelt L. Reduction of phosphorylated tau during memantine treatment of Alzheimer’s disease. Dement Geriatr Cogn Disord 2007;24(4): 247-252.
5. Reisberg B, Doody R, St?ffler A, Schmitt F, Ferris S, J?rg M?bius H. Memantine in moderate-to-severe Alzheimer’s disease. N Engl J Med 2003; 348: 1333-1341.
6. Reisberg B, Doody R, St?ffler A, Schmitt F, Ferris S, J?rg M?bius H. A 24-week open-label extension study of memantine in moderate to severe Alzheimer disease. Arch Neurol 2006; 63:49-54.
7. Chen X, Zhang ZX, Wang XD, Yao JL, Chen SD, Qian CY, et al.Multicenter research on efficacy and tolerance of memantine in Chinese patients with Alzheimer’s disease. Chinese Journal of Neurology 2007; 40(6): 364-368. (in Chinese)
8. Farlow MR. NMDA receptor antagonists. A new therapeutic approach for Alzheimer’s disease. Geriatrics 2004; 59: 22-27.
9. Bakchine S, Loft H. Memantine treatment in patients with mild to moderate Alzheimer’s disease: results of a randomized,double-blind,placebo-controlled 6-month study. J Alzheimers Dis 2007; 11(4): 471.
10. Robinson DM, Keatinq GM. Memantine: a review of its use in Alzheimer’s disease. Drugs 2006; 66(11): 1515-1534
11. Parsons CG, Danysz W, Quack G. Memantine is a clinically well tolerated N-methyl-D-aspartate (NMDA) receptor antagonist-a review of preclinical data. Neuropharmacology 1999; 38: 735-767.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition. Washington:American Psychiatric Association, 1994; pp: 147-154.
13. Folstein MF, Folstein SE, McHugh PR. Mini-mental state: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12: 189-198.
14. Hachinski VC, Lassen NA, Marshall J. Multi-infarct dementia:a cause of mental deterioration in the elderly. Lancet 1974;2(7874): 207-209.
15. Hamilton M. Development of a Psychiatric Rating Scale for Primary Depression. Brit Soc Clin Psychol 1967; 6: 278-296.
16. Rosen WG, Mohs RC, Davis KL. A new rating scale for Alzheimer’s disease. Am J Psychiatry 1984; 141: 1356-1364.
17. Lawton WP, Brody EM. Assessment of older people selfmaintaining and instrumental activities of daily living.Gerontologist 1969; 9: 179-186.
18. Guy W. ECDEU Assessment Manual for Psychopathology.Rockville, MD: U.S National Institute of Health,Psychopharmacology Research Branch; 1976: 218-221.
19. Wang LH, Shu L, Si TM, Tian CH, Zhang HY. Validity and reliability of Chinese version of Alzheimer’s Disease Assessment Scale. Chinese Journal of Clinical Psychology 2000; 8(2): 89-93. (in Chinese)
20. Li X, Xiao ZP, Xiao SF, Zhu MJ, Shen LL, Wang T, et al. Reliability and validity of Chinese version of the cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-Cog).Chinese Journal of Clinical Psychology 2009; 17(5): 538-540.(in Chinese)
21. Bullock R. Efficacy and safety of memantine in moderate to severe Alzheimer’s disease: the evidence to date. Alzheimer Dis Assoc Disord 2006; 20(1): 23-29.
22. Plosker GL, Lyseng-Williamson KA. Memantine: a pharmacoeconomic review of its use in moderate to severe Alzheimer’s disease. Pharmacoeconomics 2005; 23(2): 193-206.
23. Peskind ER, Potkin SG, Pomara N, Ott BR, Graham SM, Olin JT,et al. Memantine treatment in mild to moderate Alzheimers disease: a 24-week randomized controlled trial. Am J Geriatr Psychiatry 2006; 14: 704-715.
24. Sokusare SK, Kaul CL, Ramarao P. Dementia of Alzheimer’s disease and other neurodegenerative disorders-memantine,a new hope. Pharmacol Res 2005; 51(1): 1-17.
國產鹽酸美金剛片治療阿爾茨海默病的療效和安全性的多中心隨機雙盲平行對照研究
朱敏捷1肖世富1* 李冠軍1李霞1唐牟尼2楊斯明3許秀峰4馮連元5劉開祥6胡連平7
1上海交通大學醫學院附屬精神衛生中心,上海交通大學阿爾茨海默病診治中心 上海
2廣州市精神病醫院 廣東廣州
3郴州市第一人民醫院 湖南郴州
4昆明醫學院第一附屬醫院 云南昆明
5中國人民解放軍白求恩國際和平醫院 河北石家莊
6桂林醫學院附屬醫院 廣西桂林
7北京萬全陽光醫藥科技有限公司數據管理與統計中心 北京
背景: 鹽酸美金剛是目前用于治療阿爾茨海默病(Alzheimer's disease, AD)唯一的N -甲基- D -天門冬氨酸(N-methyl-D-aspartic acid receptor,NMDA)受體拮抗劑。目的以進口鹽酸美金剛(Ebixa)為對照,評價國產美金剛片治療中度至重度AD的有效性及安全性。方法采用隨機、雙盲雙模擬、平行對照、多中心研究方法,將229例中、重度AD患者分為研究組(國產美金剛片)和對照組(進口美金剛片),治療16周。所有的患者在基線時以及入組后第4、8、12、16周接受評估。主要療效指標為阿爾茨海默病評定量表認知分量表(Alzheimer's Disease Assessment Scale-Cognition, ADAS-Cog)評分,次要療效指標為簡易智能狀態檢查量表(Mini-Mental Statue Examination, MMSE)、日常生活能力量表(Activities of Daily Living, ADL)和臨床療效總評量表(Clinical Global Impression, CGI)的評分。結果研究組納入安全性數據集(Safety Set,SS)、全分析數據集(Full Analysis Set,FAS)和完成方案數據集(Per Protocol Set, PPS)分析的病例數分別為112、109和103例,對照組分別為111、107和101例。兩組ADAS-Cog總分和ADL總分在治療4周、8周、12周、16周末與基線相比均有所下降(p<0.001),兩組MMSE總分在治療4周、8周、12周、16周末與基線相比均有所增加(p<0.001)。各觀察時點的ADAS-Cog評分、ADL評分和病情改善程度(CGI評分)兩組之間的比較無統計學差異。研究組和對照組不良事件發生率分別為20.5%和27.0%,差異無統計學意義(χ2=1.30,p=0.255)。結論在治療中、重度AD患者的前16周中,國產美金剛片的療效和安全性與進口美金剛片相當。
Background:Memantine hydrochloride is a N-methyl-D-aspartate (NMDA) antagonist that may be useful in the treatment of Alzheimer’s disease.Aim:Compare the efficacy and safety of generic memantine hydrochloride produced in China to that of the imported proprietary version of the medication (Ebixa) in the treatment of moderate to severe Alzheimer’s disease (AD).Methods:In this multicenter, double-blind randomized controlled trial 229 patients with moderate to severe AD were randomly assigned to a 16-week trial of either the generic preparation or the proprietary preparation of memantine hydrochloride. All participants were assessed at baseline and at 4, 8, 12 and 16 weeks after enrolment. The primary outcome variable was the Alzheimer Disease Assessment Scale-cognition (ADAS-Cog)score. Secondary outcomes were scores in the Mini-Mental State Examination (MMSE), the Activities of Daily Living (ADL) scale and the Clinical Global Impression (CGI) scale.Results:Sample sizes for the safety set (SS) analysis, full analysis set (FAS) and per protocol set (PPS) analysis were 112, 109 and 103 in the generic medication group, and 111, 107 and 101 in the proprietary medication group, respectively. The ADAS-Cog and ADL total scores at the end of weeks 4, 8, 12, and 16 decreased significantly compared with baseline for both groups (p<0.001) and the MMSE total scores at the end of weeks 4, 8, 12, and 16 increased significantly compared with baseline for both groups (p<0.001). There were no significant differences in ADAS-Cog total scores, ADL total scores and level of improvement based on the CGI scores between the two groups at any of the follow-up assessments. The occurrence of adverse events was 20.5% in the generic medication group and 27.0% in the proprietary medication group; this difference was not statistically significant (χ2=1.30, p=0.255).Conclusion:There are no significant differences in the effectiveness or safety between memantine that is generically produced in China and imported proprietary memantine in the treatment of individuals with moderate and severe AD during the first 16 weeks of treatment.
10.3969/j.issn.1002-0829.2013.04.006
1Shanghai Mental Health Center, Shanghai Jiao Tong University School of Medicine; Shanghai Diagnosis and Treatment Center for Alzheimer’s disease,Shanghai, China
2Guangzhou Psychiatric Hospital, Guangzhou, Guangdong Province, China
3First People’s Hospital, Chenzhou, Hunan Province, China
4First Affiliated Hospital of Kunming Medical School, Kunming, Yunnan Province, China
5The Chinese People's Liberation Army Bethune International Peace Hospital, Shijiazhuang, Hebei Province, China
6Affiliated Hospital of Guilin Medical School, Guilin, Guangxi Province, China
7Venturepharma Center of Data Management and Analysis, Beijing, China
*correspondence: xiaoshifu@msn.com
(received: 2012-11-09; accepted:2013-01-25)

Dr. Minjie Zhu graduated from the Shanghai Second Medical University in clinical medicine in 1998 and has been working in the Shanghai Mental Health Center since then. He is currently an attending psychiatrist in the Department of Geriatric Psychiatry. His research interests include dementia, mild cognitive impairment, and geriatric mood disorders.
*通信作者:xiaoshifu@msn.com
- 上海精神醫學的其它文章
- Power analysis for cross-sectional and longitudinal study designs
- A case of recurrent neuroleptic malignant syndrome
- Should repetitive Transcranial Magnetic Stimulation (rTMS)be considered an effective adjunctive treatment for auditory hallucinations in patients with schizophrenia?
- Twelve-year retrospective analysis of outpatients with Attention-Deficit/Hyperactivity Disorder in Shanghai
- Mental health literacy among residents in Shanghai
- Prevalence of eating disorders in the general population:a systematic review

