擬諾卡氏放線菌YIM 90087 的代謝產物研究
徐 偉,丁章貴,李銘剛,文孟良,趙江源*
1云南大學云南省微生物研究所,昆明 650091;2 安徽省應用技術研究院,合肥 230031
擬諾卡氏菌屬(Nocardiopsis,Meyer 1976 年提出[1])微生物是一類高G +C 含量革蘭氏陽性好氧菌,廣泛分布于各種環境中,尤其在中度和高度鹽環境中呈優勢微生物群落。擬諾卡氏屬目前已有個49個有效描述種[2]。人們對從各種環境中分離的擬諾卡氏菌的次生代謝產物進行了研究,通過發酵分離出很多結構類型豐富的化合物。Apoptolidins A-G[3-7]、K-252 a-d[8,9]、Lucentamycin A-E[10,11]、Nocapyrones A-G[12,13]、Nocardioazine A-B[14]、Nocazines A-C、Nocazoline A[13]、Nocardiopsin A-B[15]、Naphthospironone A[16]和Griseusins F-G[17]等一系列結構新穎、具有生物活性的天然產物的發現為創新藥物研究提供了豐富的來源。
本文通過對35 株擬諾卡放線菌發酵提取物的TLC 化學多樣性進行篩選,最終將TLC 條帶最為豐富的菌株YIM90087 確定為研究菌株。該菌株是2004 年由李文均等人從中國新疆高鹽土壤中分離出來的一株擬諾卡放線菌,并鑒定為Nocardiopsis gilva sp.nov.[18]。通過對其固體發酵提取物進行化學成分研究,從中共分離得到10個化合物,這些化合物的結構類型主要為環肽類化合物,其中化合物5 為2013 年發表的一個新的三聯苯類化合物,化合物4 僅見化學合成報道,作為天然產物未見報道。
1 材料與方法
1.1 儀器與材料
Bruker AM-400 型核磁共振儀;Bruker DRX-500型核磁共振儀;Agilent G3250AA LC/MSD TOF 型質譜儀;薄層層析硅膠板和柱層析硅膠G,200~300目(青島海洋化工廠);Sephadex LH-20 葡聚糖凝膠(Amersham Pharmacia);Waters 高效液相色譜儀(515-2996);島津制備型高效液相色譜儀(LC-8A);其他化學試劑均為國產分析純。
1.2 發酵菌株
菌株YIM90087 由云南省微生物研究所唐蜀昆老師提供,該菌種于2004 年由云南省微生物研究所李文均等人鑒定為擬諾卡氏菌屬的一個新種(Nocardiopsis gilva sp.nov.),菌株保存于云南省微生物研究所。
1.3 培養基及培養條件
斜面培養基:淀粉20 g/L,蛋白胨2 g/L,酵母膏2 g/L,KNO32 g/L,MgSO40.5 g/L,K2HPO40.5 g/L,FeSO4·7H2O 0.01 g/L,1% NaCl。
種子培養基:葡萄糖10 g/L,蛋白胨10 g/L,牛肉膏5 g/L,CaCl2·2H2O 0.74 g/L,1% NaCl。
發酵培養基:葡萄糖10 g/L,蛋白胨10 g/L,牛肉膏5 g/L,CaCl2·2H2O 0.74 g/L,瓊脂15~20 g/L,pH=10,5% NaCl。
種子液制備:從新鮮斜面培養中將菌株轉接到裝有100 mL 種子培養基的500 mL 錐形瓶中,于35℃、200 rpm 搖床培養4 d。
將種子液按10%接種量轉接到固體培養基平板中,每個平板裝25 mL 培養基,共發酵1500個平板,35 ℃培養14 d。
1.4 提取與分離
將發酵好的固體培養基劃成小塊、粉碎,放于錐形瓶中用等體積丙酮超聲提取3 次,所得提取液經減壓濃縮至浸膏,將浸膏懸浮于蒸餾水后加入等體積乙酸乙酯萃取至無色,將乙酸乙酯減壓濃縮,最后得到發酵粗提物39.9 g。發酵粗提物經硅膠柱層析,用氯仿-甲醇梯度(100∶0、60∶1、40∶1、20∶1、9∶1、1∶1,V/V)洗脫,共得到9個組分(S1~S9)。S2 用石油醚-丙酮(10∶1)柱層析得到化合物5(15 mg)和6(8 mg)。S3 經凝膠Sephadex LH-20 柱層析,用甲醇洗脫得化合物1(8 mg)、2(6 mg)和3(5 mg)。S4經硅膠柱層析,用石油醚-丙酮(8∶1)洗脫得化合物4(9 mg)、7(7 mg)和8(5 mg)。S7 經硅膠柱層析,用氯仿-丙酮(9∶1)洗脫再經C18 反相柱層析,用甲醇-水(5 ∶95)洗脫得化合物9(8 mg),剩余再經HPLC 制備,用甲醇-水(10∶90)洗脫得化合物10(9 mg)。
2 結構鑒定
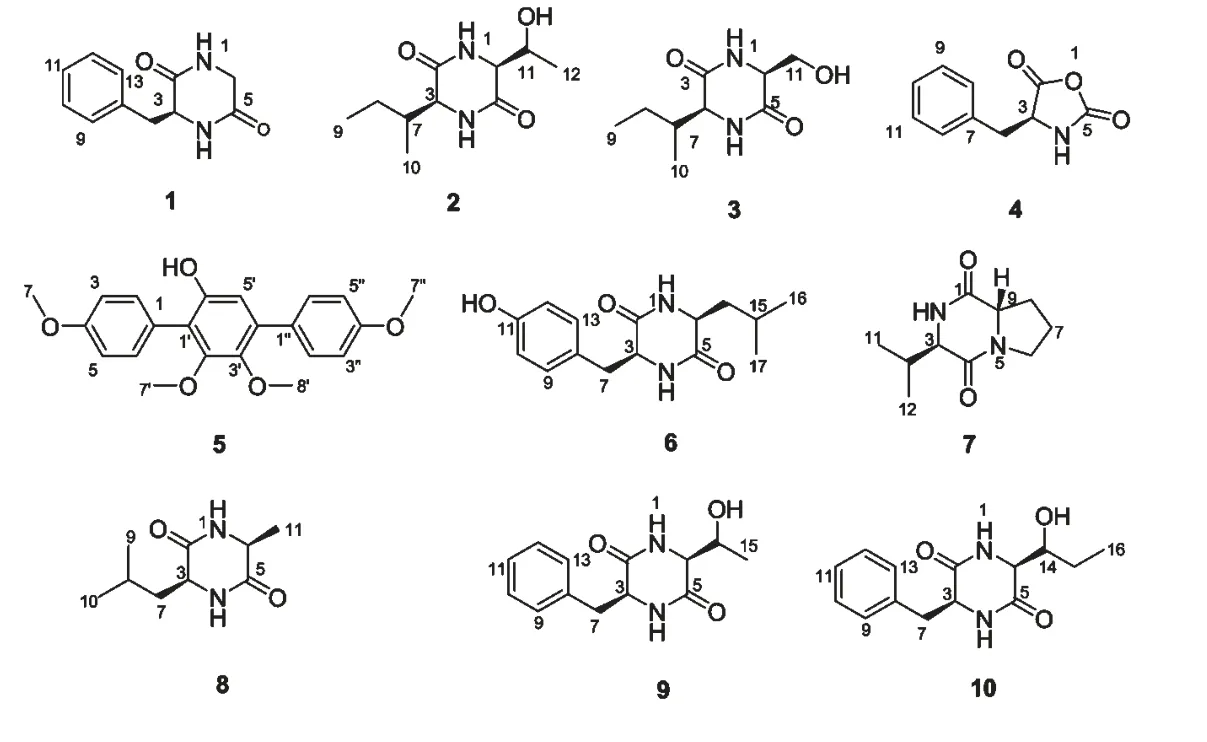
圖1 化合物1~10 的化學結構Fig.1 Chemical structures of compounds 1-10
化合物1 無色針晶(甲醇);mp.267~269 ℃;分子式為C11H12N2O2;ESI-MS m/z:227 [M +Na]+;1H NMR (DMSO,500 MHz)δ:7.92 (1H,br s,1-NH),4.06 (1H,m,H-3),8.19 (1H,br s,4-NH),3.33-2.72 (2H,d,J=17.4 Hz,H-6),3.09~2.88 (2H,dd,J=13.4,4.2 Hz,H-7),7.16 (2H,d,J=5.7 Hz,H-9,13),7.27 (2H,m,H-10,12),7.26(1H,m,H-11);13C NMR (DMSO,125 MHz)δ:167.4 (C-2),55.6 (C-3),165.9 (C-5),43.7 (C-6),38.9 (C-7),136.0 (C-8),130.2 (C-9,13),128.3 (C-10,12),126.9 (C-11)。以上數據與文獻報道[19]一致,確定化合物1 為(3S)-3-芐基-2,5-哌嗪二酮。
化合物2 白色粉末;mp.265~266 ℃;分子式為C10H18N2O3;ESI-MS m/z:237 [M+Na]+;1H NMR (DMSO,500 MHz)δ:7.89 (1H,br s,1-NH),3.57 (1H,m,H-3),8.08 (1H,br s,4-NH),3.56(1H,m,H-6),1.87 (1H,m,H-7),1.50-1.10 (2H,m,H-8),0.80 (3H,m,H-9),0.90 (3H,d,J=6.8 Hz,H-10),3.97 (1H,m,H-11),4.86 (1H,br s,11-OH),1.09 (3H,d,J=6.5 Hz,H-12);13C NMR(DMSO,125 MHz)δ:167.3 (C-2),58.9 (C-3),166.9 (C-5),60.1 (C-6),38.8(C-7),24.6(C-8),11.7(C-9),15.4(C-10),66.8(C-11),20.3(C-12)。以上數據與文獻報道[20]基本一致,確定化合物2 為(3S,6S)-3-(2-丁基)-6-(1-羥乙基)-2,5-哌嗪二酮。
化合物3 白色針晶(甲醇);mp.236~238 ℃;分子式為 C9H16N2O3;ESI-MS m/z:223 [M +Na]+;1H NMR (DMSO,500 MHz)δ:7.93 (1H,br s,1-NH),3.69 (1H,m,H-3),8.04 (1H,br s,4-NH),3.79 (1H,m,H-6),1.85 (1H,m,H-7),1.46-1.14 (2H,m,H-8),0.82 (3H,m,H-9),0.91 (3H,d,J=6.8 Hz,H-10),3.65-3.57 (2H,dd,J=10.9,2.8 Hz,H-11);13C NMR (DMSO,125 MHz)δ:166.7(C-2),58.8 (C-3),166.2 (C-5),56.8 (C-6),38.5(C-7),24.3 (C-8),11.8 (C-9),15.1 (C-10),62.5(C-11)。以上數據與文獻報道[21]基本一致,確定化合物3 為(3S,6S)-3-(2-丁基)-6-羥甲基-2,5-哌嗪二酮。
化合物4 白色針晶(甲醇);mp.90~92 ℃;[α]21D-24.5 (c 9.0,DMF);分子式為C10H9NO3;ESIMS m/z:214 [M+Na]+;1H NMR (DMSO,500 MHz)δ:4.32 (1H,m,H-3),7.93 (1H,br s,4-NH),2.92 (2H,s,H-6),7.17 (2H,d,J=7.2 Hz,H-8,12),7.26 (2H,m,H-9,11),7.21 (1H,m,H-10);13C NMR (DMSO,125 MHz)δ:175.4 (C-2),58.5(C-3),157.4 (C-5),36.5 (C-6),135.7 (C-7),129.9 (C-8,12),128.2 (C-9,11),126.8 (C-10)。以上數據與文獻報道[22]基本一致,確定化合物4 為(4S)-4-芐基-1,3-惡唑-2,5-二酮。
化合物5 白色針晶(甲醇);分子式為C22H22O5;ESI-MS m/z:389 [M +Na]+;1H NMR (CDCl3,500 MHz)δ:7.37 (1H,d,J=8.5 Hz,H-2,6),7.06(1H,d,J=8.5 Hz,H-3,5),3.88 (3H,s,H-7),6.76 (1H,s,H-5'),3.69 (3H,s,H-7'),3.61 (3H,s,H-8'),7.55 (2H,d,J=8.6 Hz,H-2'',6''),6.99(2H,d,J=8.6 Hz,H-3'',5''),3.87 (3H,s,H-7'');13C NMR (CDCl3,125 MHz)δ:124.4 (C-1),131.5 (C-2,6),114.6 (C-3,5),159.4 (C-4),55.3(C-7),121.4 (C-1'),151.4 (C-2'),144.2 (C-3'),135.4 (C-4'),111.4 (C-5'),149.1 (C-6'),60.8(C-7'),,60.7 (C-8'),130.3 (C-1''),130.2 (C-2'',6''),113.6 (C-3'',5''),158.9 (C-4''),55.3(C-7'')。以上波譜數據與文獻報道[23]一致,確定化合物5 為6'-羥基-4,2',3',4''-四甲氧基-對三聯苯。
化合物6 白色針晶(甲醇);mp.293~295 ℃;分子式為C15H20N2O3;ESI-MS m/z:299 [M +Na]+;1H NMR (DMSO,500 MHz)δ:9.15 (1H,br s,1-NH),4.66 (1H,m,H-3),9.27 (1H,br s,4-NH),4.18 (1H,m,H-6),3.57~3.24 (2H,dd,J=13.6,5.3 Hz,H-7),7.38 (2H,d,J=8.4 Hz,H-9,13),7.11 (2H,d,J=8.4 Hz,H-10,12),11.40(1H,br s,11-OH),1.65-0.97 (2H,m,H-14),1.96(1H,m,H-15),0.92 (3H,d,J=6.6 Hz,H-16),0.90 (3H,d,J=6.6 Hz,H-17);13C NMR (DMSO,125 MHz)δ:169.5 (C-2),57.7 (C-3),168.2 (C-5),54.3 (C-6),40.4 (C-7),127.7 (C-8),132.5(C-9,13),116.8 (C-10,12),158.7 (C-11),45.3(C-14),24.9 (C-15),23.7 (C-16),21.8 (C-17)。以上數據與文獻報道[24]基本一致,確定化合物6 為(3S,6S)-3-(4-羥芐基)-6-異丁基-2,5-哌嗪二酮。
化合物7 白色針晶(甲醇);mp.148~150 ℃;分子式為C10H16N2O2;ESI-MS m/z:219 [M +Na]+;1H NMR (DMSO,500 MHz)δ:5.76 (1H,br s,2-NH),3.97 (1H,m,H-3),3.63 (2H,m,H-6),2.07-1.92 (2H,m,H-7),2.41-2.05 (2H,m,H-8),4.11 (1H,d,J=8.0 Hz,H-9),2.66 (1H,m,H-10),1.09 (3H,d,J=7.7 Hz,H-11),0.95 (3H,d,J=6.8 Hz,H-12);13C NMR (DMSO,125 MHz)δ:169.9 (C-1),60.5 (C-3),165.1 (C-4),45.2 (C-6),22.4 (C-7),28.3 (C-8),58.9 (C-9),28.5 (C-10),19.5 (C-11),16.1 (C-12)。以上數據與文獻報道[25]基本一致,確定化合物7 為(3R,8aS)-3-異丙基-六氫吡咯并[1,2-a]吡嗪-1,4-二酮。
化合物8 白色針晶(甲醇);mp.230~232 ℃;分子式為 C9H16N2O2;ESI-MS m/z:207 [M +Na]+;1H NMR (DMSO,500 MHz)δ:8.11 (1H,br s,1-NH),3.76 (1H,m,H-3),8.14 (1H,br s,4-NH),3.86 (1H,dd,J=6.7,2.3Hz,H-6),1.60~1.46 (2H,m,H-7),1.81 (1H,m,H-8),0.86 (3H,d,J=6.7 Hz,H-9),0.88 (3H,d,J=6.7 Hz,H-10),1.26 (3H,d,J=6.7 Hz,H-11);13C NMR(DMSO,125 MHz)δ:168.4 (C-2),52.6 (C-3),168.9 (C-5),49.9 (C-6),42.6 (C-7),23.6 (C-8),21.9 (C-9),23.0 (C-10),19.6 (C-11)。以上數據與文獻報道[26]一致,確定化合物8 為(3S,6S)-3-異丁基-6-甲基-2,5-哌嗪二酮。
化合物9 白色針晶(甲醇);分子式為C13H16N2O3;ESI-MS m/z:271 [M +Na]+;1H NMR (DMSO,500 MHz)δ:7.88 (1H,br s,1-NH),4.08 (1H,m,H-3),8.20 (1H,br s,4-NH),3.92 (1H,d,J=3.8 Hz,H-6),2.93-2.71 (2H,dd,J=7.6,2.6 Hz,H-7),7.23 (2H,d,J=7.4 Hz,H-9,13),7.26 (2H,m,H-10,12),7.21 (1H,m,H-11),3.35 (1H,m,H-14),1.08 (3H,d,J=6.5 Hz,H-15);13C NMR(DMSO,125 MHz)δ:167.4 (C-2),56.6 (C-3),169.1 (C-5),60.9 (C-6),39.3 (C-7),136.9 (C-8),130.3 (C-9,13),128.2 (C-10,12),126.7 (C-11),72.6 (C-14),19.6 (C-15)。以上數據與文獻報道[27]基本一致,確定化合物9 為(3S,6S)-3-芐基-6-(1-羥乙基)-2,5-哌嗪二酮。
化合物10 白色針晶(甲醇);分子式為C14H18N2O3;ESI-MS m/z:285 [M +Na]+;1H NMR (DMSO,500 MHz)δ:7.79 (1H,br s,1-NH),4.06 (1H,m,H-3),8.13 (1H,br s,4-NH),3.78 (1H,d,J=3.7 Hz,H-6),2.90-2.69 (2H,dd,J=7.6,2.5 Hz,H-7),7.22 (2H,d,J=7.4 Hz,H-9,13),7.25 (2H,m,H-10,12),7.19 (1H,m,H-11),3.93 (1H,m,H-14),1.55-1.32 (2H,m,H-15),0.95 (3H,t,J=7.4 Hz,H-16);13C NMR (DMSO,125 MHz)δ:167.6(C-2),56.9 (C-3),168.3 (C-5),59.3 (C-6),39.5(C-7),136.8 (C-8),130.2 (C-9,13),128.1 (C-10,12),126.6 (C-11),73.7 (C-14),26.1 (C-15),11.3 (C-16)。以上數據與文獻報道[27]基本一致,從而確定化合物10 為(3S,6S)-3-芐基-6-(1-羥丙基)-2,5-哌嗪二酮。
3 討論
本研究從35 株擬諾卡放線菌中通過對菌株發酵提取物的TLC 化學多樣性進行篩選,最終確定了YIM90087 為研究菌株,在對目標菌株進行發酵條件優化過程中發現擬諾卡放線菌對培養基要求較高,且擬諾卡放線菌生長緩慢,由于我們對培養基的優化不夠全面,導致了發酵產物總量較少,這樣某些含量較低、成分復雜的代謝產物,不能得到有效的分離和結構鑒定,今后還需對其培養基進行更為全面的優化,并對其中的微量成分進一步深入研究。
1 Meyer J.Nocardiopsis,a new genus of the order Actinomycetales.Int J Syst Bacteriol,1976,26:487-493.
2 http://www.bacterio.cict.fr/n/nocardiopsis.html
3 Kim JW,Adachi H,Shin-ya K,et al.Apoptolidin,a new apoptosis inducer in transformed cells from Nocardiopsis sp.J Antibiot,1997,50:628-630.
4 Wender PA,Sukopp M,Longcore K.Apoptolidins B and C:isolation,structure determination,and biological activity.Org Lett,2005,7:3025-3028.
5 Wender PA,Longcore KE.Isolation,structure determination,and anti-cancer activity of Apoptolidin D.Org Lett,2007,9:691-694.
6 Wender PA,Longcore KE.Apoptolidins E and F,new glycosylated macrolactones isolated from Nocardiopsis sp.Org Lett,2009,11:5474-5477.
7 Bachmann BO,McNees R,Melancon BJ,et al.Light-induced isomerization of apoptolidin A leads to inversion of C2-C3 double bond geometry.Org Lett,2010,12:2944-2947.
8 Kase H,Iwahashi K,Matsuda Y.K-252a,a potent inhibitor of protein kinase C from microbial origin.J Antibiot,1986,39:1059-1065.
9 Nakanishi S,Matsuda Y,Iwahashi K,et al.K-252b,c and d,potent inhibitors of protein kinase C from microbial origin.J Antibiot,1986,39:1066-1072.
10 Cho JY,Williams PG,Kwon HC,et al.Lucentamycins A-D,cytotoxic peptides from the marine-derived actinomycete Nocardiopsis lucentensis.J Nat Prod,2007,70:1321-1328.
11 Cha JW,Park JS,Sim T,et al.Structure assignment of lucentamycin E and revision of the olefin geometries of the marinederived lucentamycins.J Nat Prod,2012,75:1648-1651.
12 Schneemann I,Ohlendorf B,Zinecker H,et al.Nocapyrones A-D,γ-pyrones from a Nocardiopsis strain isolated from the marine sponge Halichondria panicea.J Nat Prod,2010,73:1444-1447.
13 Fu P,Liu P,Qu H,et al.α-Pyrones and diketopiperazine derivatives from the marine-derived actinomycete Nocardiopsis dassonvillei HR10-5.J Nat Prod,2011,74:2219-2223.
14 Raju R,Piggott AM,Huang XC,et al.Nocardioazines:a novel bridged diketopiperazine scaffold from a marine-derived bacterium inhibits P-glycoprotein.Org Lett,2011,13:2770-2773.
15 Raju R,Piggott AM,Conte M,et al.Nocardiopsins:new FKBP12-binding macrolide polyketides from an australian marine-derived actinomycete,Nocardiopsis sp.Chem Eur J,2010,16:3194-3200.
16 Ding ZG(丁章貴),Li MG(李銘剛),Wen ML(文孟良),et al.Naphthospironone A:an unprecedented and highly functionalized polycyclic metabolite from an alkaline mine waste extremophile.Chem Eur J,2010,16:3902-3905.
17 Ding ZG(丁章貴),Zhao JY(趙江源),Wen ML(文孟良),et al.Griseusins F and G,spiro-naphthoquinones from a tin mine tailings-derived alkalophilic Nocardiopsis species.J Nat Prod,2012,75:1994-1998.
18 Li WJ(李文均),Kroppenstedt RM,Wang D(王棟),et al.Five novel species of the genus Nocardiopsis isolated from hypersaline soils and emended description of Nocardiopsis salina Li et al.Int J Sys Evol Microbiol,2006,56:1089-1096.
19 Gao Y(高義),Li HQ(李虎強),Zhang ZJ(張志軍),et al.Chemical constituents from an endophytic fungus Aspergillus sp.12Y03 isolated from Perilla frutescens.Acta Botanica Boreali-Occidentalia Sin(西北植物學報),2013,33:1473-1477.
20 Julia Kjer.New Natural Products from Endophytic Fungi from Mangrove Plants-Structure Elucidation and Biological Screening.Düsseldorf:Heinrich-Heine-Universit? t Düsseldorf,PhD.2009.
21 Massimo F,Michele S,Sandra C,et al.Chiral ligands containing heteroatoms.11.Optically active 2-hydroxymethyl piperazincs as catalysts in the enantioselective addition of diethylzinc to benzaldehyde.Tetra Asym,1993,4:2389-2398.
22 Miyoshi,Muneji.Peptide synthesis via N-acylated aziridinone.II.Reaction of N-acylated aziridinone and its use in peptide synthesis.Bull Chem Soc Jap.1973,46:1489-1496.
23 Tian SZ,Pu X,Luo G,et al.Isolation and characterization of new p-Terphenyls with antifungal,antibacterial,and antioxidant activities from halophilic actinomycete Nocardiopsis gilva YIM 90087.J Agric Food Chem,2013,61:3006-3012.
24 Ding ZG(丁章貴),Zhao JY(趙江源),Wen ML(文孟良),et al.1H and13C NMR assignments of eight nitrogen containing compounds from Nocardia albasp.nov(YIM 30243T).Org Magn Reson,2009,47:366-370.
25 Kwon OS,Park SH,Yun BS.Cyclo(D-Pro-L-Val),a specific β-glucosidase inhibitor produced by Aspergillus sp.F70609.J Antibiot,2001,54:179-181.
26 Wang SM(王雙明),et al.Cyclodipeptides from the roots of panax notoginseng.Nat Prot Res Dev(天然產物研究與開發),2004,16:383-386.
27 Zeng YB,Li QS,Hanzlik RP,et al.Synthesis of a small library of diketopiperazines as potential inhibitors of calpain.Bioorg Med Chem Lett,2005,15:3034-3038.

