鈦酸鋰作為鋰離子電池負極材料的改性進展
袁 華,何云蔚,艾常春
1.武漢工程大學化學與環境工程學院,湖北 武漢 430074;2.武漢工程大學化工與制藥學院,湖北 武漢 430074
鋰離子電池由于具有安全、使用壽命高、便捷可攜帶的特點使其作為一種便攜式新興能源在眾多電子產品領域得到了廣泛的應用.在鋰離子電池中,電極材料、電解質、隔膜以及其他輔助材料的研究一直是國內外新能源領域和材料學領域的研究熱點,材料科學、電化學及制造環節的突破將實現鋰離子電池的更廣泛應用.在已商品化鋰離子電池中,碳材料由于具有低倍率性能好和循環性能好的特點,是最早使用也是應用最多的負極材料,但碳材料的理論容量低(372 mAh/g),由于低電壓下易形成枝晶造成電池內部短路,從而使其大電流充放電的安全性變差[1-3],因此碳材料難以應用于動力電池等更大功率電器領域.金屬氧化物作鋰離子負極材料雖然充放電電壓略有提高,具有可逆容量高的特點,但由于充放電機理的復雜使得不可逆容量損失大,體積變化率較大,因此循環性能較差,很難實現商品化應用.氮化物作為鋰離子負極材料,具有可逆容量高,循環性能好的特點,但是制備工藝復雜,生產成本高[4-5],不利于大量的生產利用.具有尖晶石結構的鈦酸鋰作為負極材料,不僅提高了充放電電壓,而且在嵌脫鋰過程中形成的兩種物相晶格參數相近,體積效應小,被稱作零應變材料,故其安全性、循環性能及倍率性能都比碳負極材料要好,能用于大電流快速充放電.制作鈦酸鋰的材料來源廣泛,資源豐富,且對環境友好.鈦酸鋰的諸多特點,使得鈦酸鋰成為一種極富前景的負極材料.
1 結構與脫嵌鋰機理
鈦酸鋰是一種復合氧化物,由過渡金屬鈦和低電位金屬鋰組成,屬于AB2X4系,是固溶體 Li1+xTi2-xO4(0≤x≤1/3)體系中的一員,其晶體結構為尖晶石型,空間點陣群為Fd3m,晶胞參數a為0.836 nm,具有鋰離子三維擴散通道.Li4Ti5O12晶胞中,O2-位于32e位,占總數3/4的Li+位于8a的四面體間隙中,剩余的1/4的Li+和所有的Ti4+位于八面體的間隙中[6-7],其結構式可寫為[Li]8a[Li1/3Ti5/3]16d[O4]32e.
嵌入Li時,嵌入的Li和8a位的Li移到16c位,形成藍色的[Li2]16c[Li1/3Ti5/3]16d[O4]32e,嵌鋰的反應可寫為:
[Li]8a[Li1/3Ti5/3]16d[O4]32e+ Li++ e= [Li2]16c[Li1/3Ti5/3]16d[O4]32e
Li4Ti5O12的理論容量為172 mAh/g,實際容量在150~160 mAh/g[8-10],Li4Ti5O12相對金屬鋰的電位為1.55 V.
2 制備方法
鈦酸鋰的制備方法有很多,經典方法有高溫固相法、水熱法、溶膠-凝膠法,新的制備手段有靜電紡絲技術,流變相反應制備法[11]等.
Han等[12]以Li2CO3和TiO2為原料,通過0.30 mm的ZrO2高能球磨混合制得Li4Ti5O12.Chao Lai等[13]以TiOSO4·xH2O作為鈦源,以LiOH·H2O作為鋰源,使用球磨混合4 h,然后在高溫爐中500 ℃煅燒2.5 h制得多孔Li4Ti5O12, 在高電流密度10.0 C充放電時放電比容量為174.5 mAh/g,循環50圈以后放電比容量為143.4 mAh/g.在傳統的高溫固相法中,原料的混合都選用球磨的方式,在固相反應階段可以采用多種方法獲得能量[14],如高速球磨產生熱量、微波、等離子體束以及傳統的煅燒,廣泛產業化的方法還是煅燒方法.
水熱法制備鈦酸理,是將鈦源和鋰源在合適的溶劑中混合,經水熱反應得前驅體,最后高溫煅燒得鈦酸鋰[15-17].Zhang等[18]將Ti(OC4H9)4溶于乙醇,逐滴加入2 mol/L LiOH,得到懸濁液在180 ℃保存24 h得到的前驅體,再將其在500 ℃煅燒10 h制得Li4Ti5O12. 0.1 C充放電時首圈比容量為172.5 mAh/g,循環50圈后比容量為160 mAh/g,在0.5、1、10 C首圈充放電時的比容量分別為157.6、152.7、82.2 mAh/g.
溶膠凝膠法是原料在液相中混合,發生水解、縮合化學反應,在溶液中形成穩定的溶膠經陳化得到凝膠,再將凝膠干燥燒結得到材料[19-20].Zhang等[21]使用改進的溶膠凝膠法制備鈦酸鋰,EDTA和檸檬酸作螯合劑,制備出的納米級Li4Ti5O12比容量高,且倍率性能和循環性能優異.在1 C和10 C倍率下首次放電容量為164 mAh/g和104 mAh/g. 1C充放電時,在25 ℃時循環1 000次后,容量保持率高達97%.
靜電紡絲技術是Formhals在1934年提出的,至今已應用于許多的領域[22].靜電紡絲技術也能用于Li4Ti5O12材料的制備,陸海緯等[23]利用這種技術制備出鋰鹽和鈦鹽的PVP納米絲,750 ℃退火得到Li4Ti5O12納米絲三維電極. 4.5 C時首次放電容量為167.4 mAh/g,接近于0.9 C. 5次循環以后,4.5 C下的容量為138.1 mAh/g,0.9 C下為134.1 mAh/g,10次循環以后4.5 C下為114.4 mAh/g,0.9 C下為125.2 mAh/g.
在制備鈦酸鋰的方法中,水熱法和溶膠凝膠法能夠制備出形貌規整,粒度分布均勻的Li4Ti5O12材料[24](如球形納米材料),并且具有優異的電化學性能,但這些方法對設備要求高,而且產品批次不穩定,因此大規模工業化還存在一定困難.高溫固相法得到的材料形貌不如水熱法和溶膠凝膠法規整,電化學性能還有待進一步提高.靜電紡絲技術能夠得到空間結構較好的材料且性能優異,有待開發其產業化應用技術.
3 鈦酸鋰的改性
純相的鈦酸鋰,低的Li+擴散系數和低電導率(10-10S/cm)導致在大電流快速充放電時容量衰減很快,所以其改性顯得非常必要.鈦酸鋰的改性方式有納米化、形貌控制、碳包覆、金屬摻雜以及氮化處理等.
3.1 納米化及形貌控制
材料顆粒的納米化一方面可以增加電極和電解質之間的接觸面積從而提高負極材料的導電率,另一方面可以縮短Li+的外界擴散路徑,增加擴散速率,從而整體上提升材料的電化學性能. Jiang等[25]合成的Li4Ti5O12納米粒子,0.035 A/g下的放電容量為149 mAh/g,1 A/g和3 A/g放電比容量為0.035 A/g下的80%和57%,即使在更高倍率下也有較高的容量保持率.Huang等[26]使用微乳法制備出空心結構的Li4Ti5O12,該空心結構的壁是由許多100 nm的Li4Ti5O12納米粒子構建的.該材料在高倍率20 C充放電時的比容量達到95 mAh/g.在2 C下其比容量為140 mAh/g,循環500次后的比容量為135 mAh/g,每圈容量損失率僅為0.01%.將Li4Ti5O12納米粒子構建成穩固的空心球結構,不僅可以使樣品在高倍率下有較好的容量表現,而且還兼具優異的循環性能,這與空心球結構帶來的穩定性有較大關系.
Lin等[27]引入酸化碳黑合成了一種介孔Li4Ti5O12材料,圖1為該研究得到的介孔Li4Ti5O12(a) 材料和無孔Li4Ti5O12(b) 材料的SEM圖像.圖中構建球形材料的一次粒子均為納米級別,不同的是圖1a中的材料存在分布均勻的孔,Li+在材料中界面間的擴散容易得多,而無孔的材料中Li+的運輸主要是固體的體相內擴散,其速率要慢得多,因此在高倍率充放電時很難在實現Li+的快速嵌脫,不能維持穩定的容量.介孔結構的Li4Ti5O12材料在30 C下的比容量為80 mAh/g,達到了0.5 C時的57%.無孔Li4Ti5O12材料的30 C時的比容量僅為20 mAh/g.對于具有介孔結構的材料在高倍率下表現出優異的電化學性能[28],但如何保持其結構的穩定是亟待解決的問題.

圖1 介孔Li4Ti5O12材料(a)和無孔Li4Ti5O12材料(b)的圖像[26]Fig.1 The SEM images of mesoporous(a) and non-porous lithium titanate spheres(b)
控制鈦酸鋰的形貌,將材料納米化可以提高離子導電率和電子導電率,但是在循環過程中納米顆粒極易團聚,對循環穩定性帶來消極影響,因此如何獲得相對穩定的球形納米材料以及如何去固化這種結構是顆粒納米化方向的研究重點.
3.2 碳包覆
碳包覆改性方法是基于物理阻隔納米粒子的團聚以及提高顆粒材料的表面電導率而產生的,即在鈦酸理負極材料表面覆蓋一層導電物相.Luo等[29]以葡萄糖作為碳源合成了碳包覆的Li4Ti5O12.在0.2 C到30 C之間對Li4Ti5O12/C作充放電試驗,與純相的Li4Ti5O12相比,Li4Ti5O12/C表現出很好的倍率性能, 30 C下純相Li4Ti5O12的可逆容量只有46.3 mAh/g,而Li4Ti5O12/C的可逆容量高達92.7 mAh/g.Li4Ti5O12/C也表現出良好的循環性能,特別是在高倍率下,10 C充放電時,循環100次后Li4Ti5O12/C的放電容量為127.9 mAh/g,僅僅損失了3.6%,而純相Li4Ti5O12的放電容量則為95.6 mAh/g,損失7.3%.交流阻抗實驗表明Li4Ti5O12/C表現出比Li4Ti5O12低的電荷轉移阻抗(表1).也有研究采用石墨烯來包覆Li4Ti5O12材料,石墨烯不僅在一定程度上增加了Li4Ti5O12材料的導電性能,甚至其本身具有的容量還能在一定程度上增加材料的容量[30-31].
碳包覆能夠增強Li4Ti5O12材料表面的導電性,形成的碳膜阻止納米晶體的團聚使材料保持了一維納米結構,從而提升了其循環穩定性[32-33].但是對于鈦酸鋰材料,Li+擴散過程主要包括體相內的擴散、包覆層擴散和液相內擴散,其中其本身固有的離子導電率是制約其性能的關鍵因素,碳包覆對于改善其離子導電性作用具有一定局限性,因此對于該材料的體相摻雜是研究提升其離子導電率的重要途徑.
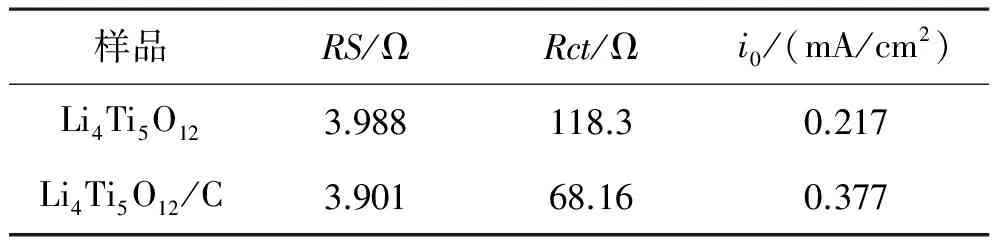
表1 純Li4Ti5O12和Li4Ti5O12/C電極的阻抗參數[29]Tabel 1 Impedance parameters of Li4Ti5O12 and Li4Ti5O12/C electrodes
3.3 金屬摻雜
體相內摻雜是采用某些其他離子替代Li4Ti5O12結構中的Li、Ti或O的位置,從而擴張Li+在體相內的傳輸路徑,達到提高離子電導率的目的.主族金屬、過渡金屬和稀土元素都可作為摻雜元素[34-39].Hu等[40]使用高溫固相法合成Li4Ti5O12和Ta摻雜后的Li4Ti4.95Ta0.05O12. Li4Ti4.95Ta0.05O12和Li4Ti5O12材料做成電池在不同電流密度下的充電實驗結果如圖2所示, Ta摻雜到Li4Ti5O12的晶體中,在低倍率下的放電比容量要比未摻雜的樣品要低,但是在高倍率下摻雜了Ta的樣品比未摻雜的樣品的首次放電容量和循環性能都要好的多.
對金屬摻雜改性鈦酸鋰,Lee等[41]對于使用不同的金屬摻雜鈦酸鋰做了比較,結果表明并不是所有的金屬摻雜都可以帶來積極的變化.制備了未被摻雜和分別用相同摩爾比的Al3+,Cr3+,Mg2+摻雜的鈦酸鋰樣品.交流阻抗的實驗和內阻測定顯示,與純的Li4Ti5O12相比,Cr3+,Mg2+的摻雜提高了Li4Ti5O12的電子導電性,而Al3+的摻雜則是降低了Li4Ti5O12的電子導電性.因此如何選擇合適的摻雜元素,在提升材料的導電性的同時又不對其晶體結構造成大的影響而影響穩定性是該項工作的關鍵.
3.4 氮化處理
氮化處理已經被較多的應用到了電化學的領域,材料經過氮化處理后能夠被活化,提高材料的表面化學反應活性.在Li-O2電池的研究中,Zhang 等[42]采用共沉淀法及氨退火處理制備出了Co3Mo3N 薄膜,該介孔納米材料和其內在的電子排布提供了許多的活性位點,使得Li-O2電池中的ORR/OER獲得優異的電催化活性.Nandi等利用Mo(CO)6和NH3得到了原子層沉積氮化薄膜[43].

圖2 Li4Ti5O12(a)和Li4Ti4.95Ta0.05O12(b)在不同電流密度下的充電實驗結果[40]Fig.2 Initial discharge-charge curves of Li4Ti5O12 (a) and Li4Ti4.95Ta0.05O12 (b) at different current densities

圖3 不同倍率下純Li4Ti5O12(A)和氮化處理的Li4Ti5O12(B)的循環性能[44]Fig.3 The cycle abilities of Li4Ti5O12 (A) and nitridated-Li4Ti5O12 (B) at different current densities.
Park等[44]將Li4Ti5O12置于NH3氛圍中,700 ℃退火得到氮化鈦酸鋰,純Li4Ti5O12和氮化處理的Li4Ti5O12的循環性能如圖3所示.在1 C以下氮化處理的Li4Ti5O12和純相的Li4Ti5O12比容量相近,但是在10 C下,氮化處理后的Li4Ti5O12的比容量高達120 mAh/g,幾乎是純相Li4Ti5O12的6倍.氮化處理后在Li4Ti5O12的表面形成了導電的TiN和Li2CO3,提高了鈦酸鋰的導電率,這種表面結構的優化使其更加適于單相Li+的嵌入和脫出.因此氮化處理可以同時提升材料的電子導電性和離子導電性,且幾乎不改變材料的晶體結構,能夠大幅提升鈦酸鋰的電化學性能,是一種較好的表面改性手段.
4 結 語
鈦酸鋰作為鋰離子電池負極材料,其制備的方法主要是高溫固相法、水熱法和溶膠凝膠法.鈦酸鋰材料的改性對于其高倍率下性能的提高意義重大,通過將材料做成納米級別可以增大材料的比表面而增大與電解質的反應面.碳材料與鈦酸鋰復合能夠改善鈦酸鋰材料的導電性,石墨烯材料與鈦酸鋰復合可以得到電化學性能不錯的復合材料.合適的金屬摻雜鈦酸鋰晶體會改善其導電性能.氮化處理沒有明顯改變鈦酸鋰的晶體結構,保持了鈦酸鋰材料零應變的特性,而能顯著改善電導率和離子導電率,得到高倍率下優異的容量性能和循環性能.在實際應用中應將幾種改性方法結合起來,采用合適的制備方法,從體相內的摻雜、表面結構的優化以及材料顆粒形貌及大小等方面綜合設計和考慮.
致 謝
感謝國家磷資源開發利用工程技術研究中心和國家級環境與化工清潔生產實驗教學示范中心和湖北省自然科學基金委員會的支持.
[1] FLANDROIS S,SIMON B. Carbon materials for lithium-ion rechargeable batteries[J].Carbon,1999,37:165-180.
[2] ENDO M,KIM C, NISHIMURA K,et al. Recent development of carbon materials for Li-ion batteries[J]. Carbon,2000,38:183-197.
[3] PROSINI P P, MANCINI R, PETRUCCI L, et al. Li4Ti5O12as anode in all-solid-state,plastic, lithium-ion batteries for low-power applications[J]. Solid State Ionics,2001,144(1):185-192.
[4] DIP K N,UTTAM K. S,DEVIKA C,et al. Atomic layer deposited molybdenum nitride thin film: a promising anode material for Li ion batteries[J]. Applied Materials Interfaces,2014,6(9):6606-6615.
[5] ZHANG Ke-jun,ZHANG Li-xue,CHEN Xiao,et al. Mesoporous cobalt molybdenum nitride: a highly active bifunctional electrocatalyst and its application in Lithium-O2batteries[J]. The Journal of Physical Chemistry C,2013,117:858?865
[6] WANG F,CHEN T,SHANG Y Z,et al. Two-phase aqueous systems of cetyltrimethylammonium bromide / sodium dodecyl sulfate with and without polyethylene glycol[J]. Korean J Chem Eng,2011,28( 3):923-926.
[7] AYDOGAN O,BAYRAKTARM E,MEHMETOGLU U,et al. Selection and optimization of an aqueous two-phase system for the recovery of 1,3-propandiol from fermentation broth[J].Eng Life Sci,2010,10( 2): 121-129.
[8] OHZUKU T,YAMAMOTO A.Zero-strain insertion material of Li4Ti5O12for rechargeable lithium cell[J]. Journal of the Electrochemical Society,1995,142(5):1431-1435.
[9] MITSUNORI K,TOMOKI A,YASUSHI M,et al. Study of surface reaction of spinel Li4Ti5O12during the first lithium insertion and extraction processes using atomic force microscopy and analytical transmission electron microscopy[J]. Langmuir,2012,7(28):12384-12392.
[10] YU Xi-qian,PAN Hui-lin,WAN Wang,et al. A size-Dependent sodium storage mechanism in Li4Ti5O12Investigated by a novel characterization technique combining in situ X-ray diffraction and chemical sodiation[J]. Nano Letters,2013,13(10):4721-4727.
[11] KATSUYA T,HIKARU I,SYOHEI T,et al. Growth of well-Developed Li4Ti5O12crystals by the cooling of a sodium chloride flux[J]. Crystal Growth Design,2011(11):4401-4405.
[12] HAN S W,RYU J H,JEONG J,et al. Solid-state synthesis of Li4Ti5O12for high power lithium ion battery[J]. Journal of Alloys and Compounds,2013,570:144-149.
[13] LAI Chao,WU Zhen-zhen,ZHU Yu-xuan,et al. Ball-milling assisted solid-state reaction synthesis of mesoporous Li4Ti5O12for lithium-ion batteries anode[J]. Journal of Power Sources,2013,226:71-74.
[14] HAN J,WANG Y,YU C L,et al. Tetrafluoroborate and ammonium tartrate at different temperatures: experimental determination and correlation [J].Anal Bioanal Chem,2011,399: 1295-1304.
[15] YAN Hui, ZHU Zhi, ZHANG Ding,et al. A new hydrothermal synthesis of spherical Li4Ti5O12anode material for lithium-ion secondary batteries[J]. Journal of Power Sources, 2012,219:45-51.
[16] LIU Jian,LI Xi-fei,YANG Jin-li,et al. Microwave-assisted hydrothermal synthesis of nanostructured spinel Li4Ti5O12as anode materials for lithium ion batteries[J]. Electrochimica Acta,2012,63:100-104.
[17] RAHMAN M M,WANG J Z,HASSAN M F,et al. Basic molten salt process -A new route for synthesis of nanocrystalline Li4Ti5O12-TiO2anode material for Li -ion batteries using eutectic mixture of LiNO3-LiOH -Li2O2[J]. Journal of Power Sources,2010,195( 13):4297 -4303.
[18] ZHANG Zhen-wei,CAO Li-yun,HUANG Jian-feng,et al. Hydrothermal synthesis of Li4Ti5O12microsphere with high capacity as anode material for lithium ion batteries[J]. Ceramics International, 2013,39:2695-2698.
[19] FANG Wei,CHENG Xin-qun ,ZUO Peng-jian ,et al. Hydrothermal-assisted sol-gel synthesis of Li4Ti5O12/C nano-composite for high-energy lithium-ion batteries[J]. Solid State Ionics,2014,244:52-56.
[20] LEE S B,JANG I C,LIM H H,et al. Preparation and electrochemical characterization of LiFePO4nanoparticles with high rate capability by a sol-gel method[J]. J Alloy Compd,2010,491(12):668-672.
[21] ZHANG Chun-ming,ZHANG Yao-yao,WANG Jin,et al. Li4Ti5O12prepared by a modified citric acid solegel method for lithium-ion Battery[J]. Journal of Power Sources,2013,236:118-125.
[22] MIAO Yue-e, FAN Wei, CHEN Dan, et al.High-performance supercapacitors based on hollow polyaniline nanofibers by electrospinning[J]. ACS Applied Materials & Iinterfaces,2013,5(10):4423-4428.
[23] 陸海緯,周永寧,李越生,等. 三維尖晶石Li4Ti5O12納米絲網狀帶電極的構置與化學性能,無機化學學報,2006,5(2):1802-1806.
LU Hai-wei, ZHOU Yong-ning, LI Yue-sheng et al. Fabrication and electrochemical properties of 3-dimentional net architectures of spinal Li4Ti5O12nanofibers[J]. Chinese Journal of Inorganic Chemistry, 2006,5(2):1802-1806 .(in Chinese)
[24] ABDELFATTAH M,JOSE M A,KARIMA L,et al. Influence of the synthesis method on the electrochemical properties of the Li4Ti5O12spinel in Li-half and Li-ion full-cells. A systematic comparison[J]. Electrochimica Acta,2013,93:163-172.
[25] JIANG Chun-hai,EIJI H, MASKI I,et al. Synthesis of nanocrystalline Li4Ti5O12by chemical lithiation of anatase nanocrystals and postannealing[J]. Journal of The Electrochemical Society,2008,155(8):553-556.
[26] HUANG Jun-jie ,JIANG Zhi-yu. The Synthesis of hollow spherical Li4Ti5O12by macroemulsion method and its application in Li-ion batteries[J]. Electrochemical and Solid-State Letters,2008,11(7):116-118.
[27] LIN Yu-sheng,DUH Jenq-gong. Facile synthesis of mesoporous lithium titanate spheres for high rate lithium-ion batteries[J]. Journal of Power Sources,2011,196:10698-10703.
[28] LI Cheng-chao,LI Qiu-hong,CHEN Li-bao,et al. A facile titanium glycolate precursor route to mesoporous Au/Li4Ti5O12spheres for high-Rate lithium-ion batteries[J]. Applied Materials Interfaces,2012, 4:1233-1238.
[29] LUO Hong-jun,SHEN Lai-fa,RUI Kun ,et al. Carbon coated Li4Ti5O12nanorods as superior anode material for high rate lithium ion batteries[J]. Journal of Alloys and Compounds,2013,572:37-42.
[30] HAO Shuai,XIAO Xiao-ling,HU Zhong-bo,et al. Improving the electrochemical performance of Li4Ti5O12anode through confinement into ordered bimodal porous carbon frameworks[J]. The Journal of Physical Chemistry C, 2013, 117 (51):26889-26895.
[31] ZHU N,LIU W,XUE M,et al. Graphene as a conductive additive to enhance the high rate capabilities of electrospun Li4Ti5O12for lithium -ion batteries [J]. Electrochimical Acta,2010,55( 20): 5813 -5818.
[32] RAI A K,GIM J,KANG Sung-Won,et al. Improved electrochemical performance of Li4Ti5O12with a variable amount of graphene as a conductive agent for rechargeable lithium-ion batteries by solvothermal method[J]. Materials Chemistry and Physics,2012,136:1044-1051.
[33] CHENG L,YAN J,ZHU G N,et al. General synthesis of carbon coated nanostructure Li4Ti5O12as a high rate electrode material for Li -ion intercalation [J]. Journal of Material Chemistry,2010,20 ( 3 ):595-602.
[34] WANG Zhen-hong, CHEN Gang. Synthesis and electrochemical performances of Li4Ti4.95Al0.05O12/C as anode material for lithium-ion batteries[J]. Journal of Physics and Chemistry of Solids, 2011,72(6):773-778.
[35] ZHAO Hai-lei, LI Yue, ZHU Zhi-ming. Structural and electrochemical characteristics of Li4-xAlxTi5O12as anode material for lithium-ion batteries[J]. Electrochimica Acta. 2008,53:7079-7083.
[36] TIAN Bing-bing,XIANG Hong-fa,ZHANG Le,et al. Niobium doped lithium titanate as a high rate anode material for Li-ion batteries[J]. Electrochimica Acta,2010,55:5453-5458.
[37] SHARMILA S,SENTHILKUMARB,NITHYA V D,et al. Electrical and electrochemical properties of moltensalt-synthesized Li4Ti5-xSnxO12(x1/40.0, 0.05and0.1)asanodesforLi-ionbatteries[J]. Journal of Physics and Chemistry of Solids,2013,74:1515-1523.
[38] JI Shuang-ze, ZHANG Jun-ying. Preparation and effects of Mg-doping on the electrochemical properties of spinel Li4Ti5O12as anode material for lithium-ion battery[J]. Materials Chemistry and Physics,2010,123:510-515.
[39] ARUMUGAM S, SUKUMARAN G. Novel Li4Ti5O12/Sn nano-composites as anode material for lithium ion batteries[J]. Materials Research Bulletin, 2011,46:492-500.
[40] HU Guo-rong,ZHANG Xin-long,PENG Zhong-dong. Preparation and electrochemical performance of tantalum-doped lithium titanate as anode material for lithium-ion battery[J]. Transactions of Nonferrous Metals Society of China,2011,21:2248-2253.
[41] LEE B,YOON J R. Preparation and characteristics of Li4Ti5O12with various dopants as anode electrode for hybrid supercapacitor[J]. Current Applied Physics,2013,13:1350-1353.
[42] ZHANG Ke-jun,ZHANG Li-xue,CHEN Xiao,et al. Mesoporous cobalt molybdenum nitride: a highly active bifunctional electrocatalyst and its application in lithium-O2batteries[J]. Jounrnal of Physical Chemistry C,2013,117:858-865.
[43] NANDI D K.,SEN U K.,DEVIKA C,et al. Atomic layer deposited molybdenum nitride thin film: a promising anode material for Li ion batteries[J]. ACS Applied Materials & Interfaces,2014:6606-6615.
[44] PARK Kyu-Sung,BENAYAD A,KANG Dae-Joon,et al. Nitridation-driven conductive Li4Ti5O12for lithium ion batteries[J]. Journal of the American Chemical Society,2008,130:14930-14931.

