畜禽養殖場顆粒物污染特征及其危害呼吸道健康的研究進展
戴鵬遠,沈丹,唐倩,李延森,李春梅
?
畜禽養殖場顆粒物污染特征及其危害呼吸道健康的研究進展
戴鵬遠,沈丹,唐倩,李延森,李春梅
(南京農業大學動物科技學院,南京 210095)
隨著畜禽養殖集約化程度的提高,高密度飼養引起畜禽養殖場空氣質量問題日益突出,特別是養殖舍內環境顆粒物(particulate matter:PM)污染引起的家畜呼吸道健康問題不容忽視。畜禽養殖生產過程中可產生大量PM,已成為大氣細顆粒物PM2.5(空氣動力學直徑小于等于2.5 μm)和PM10(空氣動力學直徑小于等于10 μm)的重要來源,影響大氣環境空氣質量。畜禽養殖場的PM主要來源于飼料、糞便、羽毛、皮屑等,其成分主要是有機物,含有C、H、O、N、S、Ca、Na、Mg、Al和K等多種元素;PM表面還附著細菌、真菌、病毒等多種微生物以及內毒素、氨氣、硫化氫等有害物質。畜禽養殖舍PM的產生和釋放受到家畜的種類、日齡、活動以及季節等多種因素的影響,雞舍內PM的濃度高于豬舍,冬季舍內PM的濃度高于夏季。但是,目前缺少標準化設備和標準方法來測量不同類型的畜禽舍PM的濃度和排放水平。畜禽養殖舍PM的成分復雜,具有很強的生物學效應,嚴重危害家畜的健康和生產。畜禽舍內高濃度PM主要通過以下3種形式影響呼吸道健康,一是PM直接刺激呼吸道,降低機體對呼吸系統疾病的免疫抵制;其次是PM表面附著的多種化合物的刺激;第三種是PM表面的病原性和非病原性微生物的刺激。目前關于PM對呼吸道健康危害機制的研究主要集中在PM對呼吸道的致炎作用,研究發現:PM通過刺激肺泡巨噬細胞產生前炎癥因子,繼而誘發其它細胞釋放炎癥因子,引起肺發生炎癥反應;另外,PM2.5通過引起肺組織細胞發生氧化應激,激活絲裂原活化蛋白激酶 (MAPKs)活性,上調核轉錄因子κB (NFκB) 和轉錄激活因子AP-1的表達而誘發肺的炎癥; PM2.5也可通過激活模式識別受體Toll樣受體TLR2和TLR4的表達,激活NFκB信號通路而導致炎癥的發生。也有研究發現,PM2.5在誘導呼吸道炎癥的同時,還會激活細胞自噬和核因子相關因子-2(nuclear factor E2-related factor 2,Nrf2)相關信號通路,這為緩解和治療PM引起細胞損傷提供了靶點。盡管PM危害呼吸道健康的機制研究較多,但是PM成分復雜,并處在不斷變化中,因此PM誘導呼吸道損傷的機制也十分復雜,仍需進一步系統深入研究。畜禽養殖生產過程中釋放的大量PM嚴重影響環境空氣質量和家畜健康,而PM對環境和家畜健康的危害程度與其組成和濃度密切相關。因此,正確認識畜禽舍PM的形態、大小、組成、濃度水平及其形成排放影響因素,對確定畜禽舍PM的來源和PM的毒性危害具有重要意義。文章就畜禽生產過程中產生的PM的來源、化學組成、濃度、排放、影響因素,以及PM對呼吸道功能的影響及作用機制作一綜述,為正確評估PM對畜禽健康生產的影響提供參考依據。
畜禽舍; 顆粒物; 污染特征;呼吸道危害
畜禽生產過程中產生并釋放大量的顆粒物(particulate matter,PM)對家畜的健康和生產以及現場工作人員健康產生不利影響。PM通過呼吸進入呼吸道,嚴重危害動物和人的呼吸道健康[1,2]。長期暴露于PM2.5濃度較高雞舍的生產管理一線人員,易患呼吸道疾病、哮喘以及慢性阻塞性肺病[3]。另外,舍內PM通過通風設施排放到大氣中,還會對大氣造成污染。有研究表明大氣中PM10和PM2.5的8%和4%來自于畜禽生產[4-5]。據報道,荷蘭大氣中PM的25%來自于農業[6];歐洲集約化的禽舍和豬舍是大氣PM的重要來源,分別占到50%和30%[7]。高濃度的PM除了影響大氣環境,還會影響云的形成、大氣可見度等[8]。畜禽舍PM表面附著氨氣、惡臭化合物及微生物等,這些物質增強了PM的毒性。因此,正確認識畜禽舍環境中PM的形態、大小、組成、濃度水平及其形成和排放影響因素,對確定畜禽舍PM的來源和PM的毒性危害具有重要意義。本文針對畜禽生產過程中產生的PM的來源、化學組成、濃度、排放、影響因素,以及PM對呼吸道功能的影響及作用機制作一總結,為正確評估PM對畜禽健康生產的影響提供參考依據。
1 PM的基本特征和分類
大氣PM一般是指粒徑小的、分散的、懸浮在氣態介質中的固體或氣體粒子。大氣PM一般可分為初級PM和次級PM。初級PM是直接釋放到大氣中的粒子,包括土壤粒子、海鹽粒子、生物碎片等;次級PM是指大氣中污染氣體組分與正常氣體組分通過化學反應生成的PM。根據PM的形成和來源不同,其性質、形狀、大小、密度和化學組成也不盡相同,這就造成了PM的異質性[9]。這種異質性同樣適用于畜禽舍中的PM。圖1是雞舍中PM2.5電鏡圖片,顆粒物呈現出薄片狀、橢圓形以及晶體狀,直徑在幾納米到十微米之間[10]。畜禽舍內的PM濃度一般為其它室內PM濃度的10—100倍,它是多種氣味化合物及氨氣、硫化氫等氣體的載體,其表面一般附著多種不同種類的微生物[11]。
通常,采用空氣動力學當量直徑(aerodynamic equivalent diameter, AED)來描述大氣粒子大小。不論粒子形狀、大小和密度如何,當它與密度為1 g·cm-3球體粒子沉降速度一致時,該球體的直徑就是該粒子的直徑。對于不規則形狀的顆粒物,這個直徑是一個有用的測量指數,因為具有相同AED的粒子懸浮在空氣中的行為表現可能相同[12]。
顆粒物的分類方法主要有沉降特性法、粒子大小法和健康大小法。按照沉降特性法,顆粒物分為降塵和飄塵。降塵一般是指粒徑大于10 μm的粒子,它們在空中易于沉降,速度大約為 0.3 cm /s,當粒子直徑大于30 μm時,沉降速度為1 cm /s。飄塵是指粒徑小于10 μm,能在空氣中長期漂浮的粒子。根據粒子大小法,顆粒物可分為總懸浮顆粒物(total suspended particulates, TSP,粒子直徑0—100mm),粗顆粒物(粒子直徑介于2.5—10 μm,PM2.5-10),細顆粒物(粒子直徑小于 2.5 μm,PM2.5)和超細顆粒物(粒子直徑小于0.1 μm,PM0.1)。健康大小法是根據顆粒物進入呼吸道不同深度來進行分類的。國際標準化組織規定將直徑小于等于10 μm 的顆粒物定為可吸入顆粒物,在可吸入顆粒物中,大于5 μm的粒子被阻擋在上呼吸道,小于5 μm的粒子進入氣管和支氣管,而粒徑小于2.5 μm的粒子能進入肺泡,這部分顆粒物稱為可呼吸顆粒[13]。

圖1 雞舍中不同來源PM的掃描電鏡圖片
2 畜禽舍PM的特征
2.1 畜禽舍PM的來源
有關畜禽舍PM來源的相關研究已有較多報道,主要集中在豬舍和雞舍。豬舍中的PM主要來源于飼料和糞便[14-15],育肥豬舍中的PM含有較高濃度的氮,這表明飼料、動物皮屑和其它含氮化合物為舍內PM的主要來源[16]。另外,霉菌、谷物、昆蟲以及礦物質粉塵也是豬舍PM的來源[14]。根據顆粒物大小的源解析表明,豬舍中PM的5%—10%來源于皮膚,粒徑在7—9 μm的粒子占5%,粒徑為11—16 μm的粒徑占10%[17]。在肉雞舍中,PM的主要來源為雞絨羽、尿中的礦物晶體以及廢棄物[18]。蛋雞舍PM的主要來源包括皮屑、尿液、飼料及廢棄物[19]。另外,與無墊料雞舍相比,有墊料雞舍里墊料也是PM的主要來源[20-21]。在生長育肥豬舍內,含有稻草墊料豬舍比混凝土地板豬舍PM濃度高兩倍,尤其在育肥后期舍內PM濃度更高,這是因為在后期,墊料更臟,易被分解產生更多的顆粒物[20]。
2.2 畜禽舍PM的化學組成
畜禽舍內90%的PM由有機粒子組成[22],主要有生物來源的初級粒子,如真菌、細菌、病毒、內毒素及過敏原,還有來源于飼料、皮膚和糞便的粒子等[14]。舍內PM的組成成分與家畜種類、畜禽舍廢棄物(畜禽糞便、畜禽舍墊料、廢飼料及散落的毛羽等廢物)的組成有關[23-24]。畜禽舍PM成分中主要的元素為C、O、N、P、S、Na、Ca、Al、Mg和K[15, 25]。豬舍和禽舍內的PM富含N元素,而來自于牛舍的PM中N元素含量少。牛舍中PM濕度較大同時含有較多的礦物質和灰燼[26]。對育肥豬舍PM成分分析結果發現,Na、Mg、Al、P、S、Cl、K及Ca 含量較高[20]。也有研究報道,豬舍內不同粒徑的PM中P、N、K及Ca含量較高[27]。糞便粒子含有較高的C、P和較高的有機磷酸酯和焦磷酸鹽[28]。在肉雞舍中不同來源的PM2.5和PM10中含有的元素成分不同,糞便來源的PM中N,Mg,P和K元素含量最高;皮膚來源的PM中S元素含量最高;木屑來源的PM中Na和Cl元素濃度最高;飼料來源的PM中Si和Ca元素含量最高;舍外的PM2.5中Al元素含量最高[29]。
2.3 畜禽舍PM的濃度、排放及其影響因素
畜禽舍內PM的濃度取決于多種因素,包括家畜的種類、飼養方式、活動情況、飼養密度以及舍內環控系統、舍內濕度、季節及采樣時間等[25,30]。豬舍內PM的濃度和排放與舍內的通風率、濕度及豬的活動量、飼養管理、體重及育肥狀態有關[31,32]。多因子線性分析揭示了在肉雞舍中,通風效率、墊料類型、舍內溫度、建筑物年限對舍內PM10的濃度影響較大,而舍內PM2.5的濃度與舍內雞數量、通風水平及濕度有關[33]。通風率,溫度和相對濕度是影響PM形成的重要因素,它們決定了PM的形成、排放過程和粒子分布[25,34-35]。在肉雞舍中的研究結果表明,舍內溫度和相對濕度對TSP濃度影響較大[18, 36]。
2.3.1 不同家畜種類對舍內PM濃度的影響 CAMBRA-LóPEZ 等[37]對不同畜舍內PM濃度進行監測分析對比發現,禽舍中PM的濃度高于豬舍,肉雞舍PM濃度高于籠養蛋雞舍,火雞舍中TSP濃度為1.3—7.5 mg·cm-3,與肉雞舍中TSP濃度相近。牛舍中PM濃度相對較低,奶牛舍中TSP濃度低于1 mg·cm-3[38-39]。表1和表2分別總結了禽舍和豬舍中TSP和PM4監測到的濃度范圍。禽舍中的PM濃度高于豬舍,肉雞舍中的PM濃度高于蛋雞舍,平養蛋雞舍PM濃度高于籠養蛋雞舍。
2.3.2 家畜日齡對舍內PM濃度的影響 有研究報道,肉雞舍PM的濃度隨著肉雞日齡的增加呈線性增長[40]。YODER等[41]也研究發現禽舍PM2.5的濃度隨動物日齡的增加呈現對數增長。禽類日齡增加引起舍內PM濃度升高可能是因為隨著動物日齡的增長,干糞增多,雞活動增強,羽毛量增多等原因。相反,豬舍內PM10濃度隨著豬體重的增加而降低,這可能是因為隨著體重增加,豬的活動量減少的原因[40]。
2.3.3 家畜活動對舍內PM濃度的影響 每天的喂料時間以及光照程序會通過影響家畜活動來影響舍內PM的形成和濃度。白天,由于喂料和養殖人員的活動,家畜采食等活動量增多,易引起畜舍建筑物表面的PM分散,使舍內PM濃度上升[42]。相對于肉雞,蛋雞在白天的活動量較大,因此蛋雞舍內PM的濃度相對較高[42-43]。另外,光照也會影響畜禽舍內PM濃度的變化。在雞舍中,光照較強位置處的PM濃度明顯高于黑暗處,這是因為在光照下動物活動增強[19]。在間歇性光照程序的肉雞舍中,舍內PM濃度與光照時間及動物活動具有很強的關系。在豬舍中,PM10在喂料時間明顯升高,白天PM濃度高于夜間[32,40]。這些結果充分說明,動物活動與PM濃度密切相關,任何能引起動物活動的因素均會影響PM的濃度和分布。
2.3.4 季節因素對舍內PM濃度的影響 季節因素引起的PM濃度變化與舍內通風率密切相關[44]。增加通風率可降低畜禽舍內PM濃度,夏季舍內的通風率比冬季高,因此夏季舍內PM的排放率較高,舍內PM濃度較低[44]。研究表明,不論雞舍還是豬舍,夏季的舍內PM濃度低于冬季[42]。HINZ等[40]研究發現,當夏季舍內外溫差小于10℃時,肉雞舍內PM的濃度低于冬季舍內外溫差大于10℃時的濃度。舍內外較小的溫差與高通風率相關,這些進一步說明通風率對PM濃度具有很大影響。
3 畜禽舍PM對呼吸道健康的危害及其可能機制
3.1 畜禽舍PM對呼吸道健康危害
畜禽舍內高濃度PM容易引起家畜呼吸道疾病[53-54]。PM通過以下3種方式影響呼吸道健康,第一種是PM直接刺激呼吸道,降低機體對呼吸系統疾病的免疫抵制;其次是PM表面附著的化合物的刺激;第三種是PM表面病原性和非病原性微生物的刺激[55]。第一種方式是顆粒物本身引起的呼吸道損傷[53, 56],此種方式同時與第二和第三種方式相關[57-58]。畜禽舍PM的表面附著大量的重金屬離子、揮發性有機化合物(volatile organic chemicals, VOCs)、NO3-、SO42-、NH3、臭味化合物、內毒素、抗生素、過敏原、塵螨及β-葡聚糖等物質[59-60],這些物質以PM為載體進一步危害呼吸道健康。PM影響呼吸道健康的第三種方式與生物氣溶膠相關,PM表面附著的大量細菌、真菌和內毒素,易引起呼吸道感染[61- 62]。畜禽舍空氣中革蘭氏陰性菌所占比例盡管低于10%,但所有的革蘭氏陰性菌均具有致病性[63]。內毒素是革蘭氏陰性菌細胞膜中的脂多糖成分,在畜禽舍周圍內毒素的濃度高達0.66—23.22 EU(endotoxin units)/m3[64],在牛舍中內毒素濃度最高可達761 EU/m3[65],散養蛋雞舍的內毒素最高濃度可達8120 EU/m3[66]。這些高濃度的內毒素不僅可引起家畜呼吸道和肺部感染,同時也危害養殖工作人員及其周邊居民的呼吸道健康[67-68]。致病性生物氣溶膠不僅可以直接損害家畜呼吸道健康,還可以通過空氣傳播擴散到鄰近農場[52]。
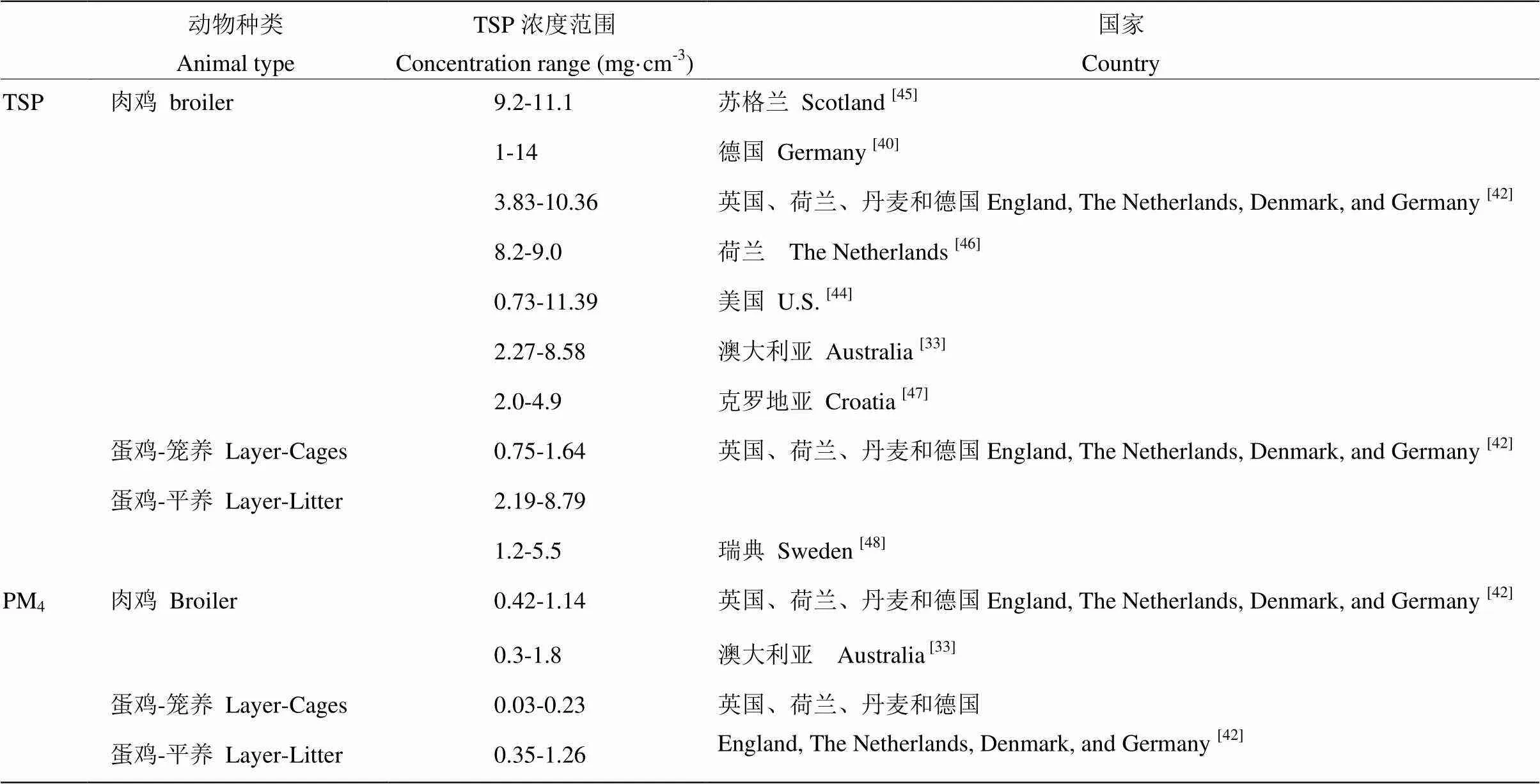
表1 肉雞舍和蛋雞舍內TSP和PM4的濃度

表2 豬舍內TSP和PM4的濃度
有關畜禽舍PM損害呼吸道功能的研究已有很多文獻報道,但這些研究多集中在體外試驗研究。有研究報道,豬場內的顆粒粉塵導致人類肺泡巨噬細胞和支氣管上皮細胞的炎癥因子白細胞介素-8(IL-8)、白介素-12(IL-12)和IFN-γ表達上調,提高活化T細胞的百分率[69],并增強對呼吸感染的易感性[70]。另外,近年來有些研究者對豬舍內有機灰塵的提取物(organic dust extract,ODE)進行的體內和體外試驗研究發現,ODE不僅能引起豬呼吸道和肺部的炎癥,導致肺部細胞發生凋亡[71],增加炎癥細胞數[72-73],還可降低炎癥細胞的吞噬能力以及細菌殺傷能力[74],引起呼吸道的高反應性[75]。MITLOEHNER等[76]研究發現奶牛場PM可降低奶牛場工作人員的肺活量,這與亞急性呼吸道損害相關。
3.2 畜禽舍PM誘導呼吸道炎癥反應機制
3.2.1 PM誘導肺泡細胞炎癥反應的機制 PM引起的呼吸道危害主要與肺部炎癥相關,因此下面集中討論PM誘導的肺炎癥反應的相關機制。吸入的PM能刺激肺泡巨噬細胞產生前炎癥因子,前炎癥因子刺激肺泡的上皮細胞、內皮細胞及成纖維母細胞分泌細胞因子和黏附因子,誘導炎性細胞聚集,引發炎癥反應[77]。PM誘導炎癥反應的一個重要機制是氧化應激[78],氧化應激是活性氧(reactive oxygen species, ROS)的產生與抗氧化體系不平衡所造成的[79]。PM能刺激機體呼吸道組織細胞產生ROS,而ROS能激活氧化還原敏感性信號轉導通路,如絲裂原活化蛋白激酶(MAPKs)和磷脂酰肌醇-3-激酶/蛋白激酶B (PI3K/AKT)通路[80-82]。MAPKs包含一組絲氨酸/蘇氨酸蛋白激酶(c-Jun NH2-末端激酶,JNK;胞外信號調節激酶,ERKs;應激激活蛋白激酶,p38),它們能在細胞外應激源的刺激下被激活,調節從細胞表面到核的信號轉導,最終導致致炎因子的表達上調而引起細胞炎癥反應[83]。有研究報道,PM可誘導人和鼠的肺泡巨噬細胞產生過多的ROS,進而激活MAPKs,誘導轉錄激活因子AP-1的表達上調,誘發細胞炎癥反應[84]。柴油機廢氣粒子能誘導人氣管上皮細胞產生ROS,激活ERK1/2和p38,繼而激活下游的核轉錄因子κB(NFκB), 最終誘發細胞發生炎癥反應[85]。鈣離子(Ca2+)是維持生命活動不可缺少的離子,其在凝血、肌肉收縮、神經遞質的合成與釋放、機體免疫功能方面發揮重要的作用[86-87]。研究發現,PM引起肺泡上皮細胞的氧化應激刺激Ca2+從細胞內質網中釋放出來,調節轉錄因子NFκB的表達,促進炎癥因子表達的上調[88]。
PM誘導細胞炎癥反應的另一機制是通過Toll樣受體(TLRs)信號通路[89-90]。TLRs是一種模式識別受體(pattern recognition receptor, PRR),表達于固有免疫細胞表面,能識別一種或多種病原體相關分子模式(pathogen-associated molecular patterns, PAMP), 在先天性免疫和獲得性免疫系統中發揮作用,目前發現TLR受體共有13種,包括TLR1-13[91-92]。大量研究發現,大氣粒子污染物能激活細胞模式識別受體TLR2和TLR4[93-95]。髓樣分化因子88(MyD88)和TIR 結構域銜接蛋白(TRIF)是粒子暴露引起表達的潛在下游蛋白,它是所有TLRs的接頭蛋白[96-97]。以小鼠為模型急性感染ODE引起的炎癥反應主要是通過MyD88信號通路[98]。肺巨噬細胞在PM的刺激下,TLR4與TRIF 相關接頭分子(trif-related adaptor molecule,TRAM)結合招募 TRIF,進而激活p38, 引起下游炎癥因子表達上調,最終導致細胞發生炎癥反應[84]。
3.2.2 PM誘導呼吸道損傷的可能緩解機制 自噬是一種進化上保守并且與溶酶體信號通路相關的過程,它通過多重通路降解蛋白質、糖原、脂質、核苷酸等大分子以及細胞器[99]。自噬被證明參與許多生理過程,包括宿主防御,細胞生存和死亡[100]。近來研究發現PM暴露可誘導細胞自噬發生。鄧等[101]利用腺癌人類肺泡Ⅱ型上皮細胞(A549細胞)研究發現,PM2.5可誘導A549細胞自噬標志蛋白LC3Ⅱ以一種時間和濃度依賴的方式累積,并且促進自噬起始蛋白Beclin1和相關基因Atg5的高表達。PM2.5暴露大鼠肺巨噬細胞和小鼠腹腔巨噬細胞,可誘導細胞ROS產生,激活PI3K/AKT信號通路,同時降低自噬中心蛋白—哺乳動物雷帕霉素靶蛋白(mammalian target of rapamycin, mTOR)的表達,增強了LC3Ⅱ的產生[102-103]。AMP依賴的蛋白激酶(AMPK)負責細胞能量代謝,它能被外界各種應激源刺激激活,以保持機體葡萄糖代謝平衡。另有研究發現,PM2.5可激活A549細胞AMPK的表達,進而抑制mTOR的表達,同時促進自噬核心蛋白ULK1的表達[104]。細胞自噬和炎癥反應關系十分密切。細胞在PAMP刺激下引發炎癥和自噬,而自噬對炎癥具有負調節作用[105]。自噬抑制炎癥發生的機制可能是通過降低細胞內ROS水平實現的。自噬能抑制細胞內線粒體的聚集,及時清除胞內的過氧化物酶體,進而減少由ROS誘導的炎癥反應[106-107]。
核因子相關因子-2(nuclear factor E2-related factor 2,Nrf2)是一種轉錄因子,細胞在正常狀態下,Nrf2與Keap1結合被錨定胞質,應激發生時,Keap1被降解,Nrf2解離進入細胞核與抗氧化反應原件(antioxidant response element, ARE)結合,進而啟動下游抗氧化基因的轉錄表達。細胞在外界應激源刺激下產生ROS,使細胞發生氧化應激,而過量的ROS又激活了Nrf2抗氧化信號通路,從而減輕細胞的氧化損傷。PM2.5暴露A549細胞,可誘導細胞產生ROS, 而ROS激活Nrf2抗氧化信號通路,從而減輕PM2.5對細胞的毒性損傷。除了PM刺激,研究證明Nrf2信號通路在多種肺部炎癥性疾病中發揮作用[108]。當肺部組織受到有毒有害物質刺激時,Nrf2信號通路能被激活,上調抗氧化基因的表達,進而減輕因應激因素造成的肺損傷。此外,也有研究報道,急性肺炎治療的藥物主要通過上調細胞Nrf2的表達,抑制NFκB和AP-1生成,降低炎癥因子表達而最終起到消炎作用[109]。綜合上述,用圖4總結概述PM誘導細胞炎癥反應、自噬和Nrf2信號通路的可能機制。

圖2 PM誘導細胞炎癥反應、自噬和Nrf2信號通路的可能機制
4 結語和展望
4.1 畜禽養殖生產過程中釋放的大量顆粒物嚴重影響環境空氣質量和家畜健康,而顆粒物對環境和家畜健康的危害程度與其組成和濃度密切相關,因而確切掌握顆粒物的組成和形態以及排放規律對減少和控制顆粒物的排放非常重要。然而,目前畜禽舍顆粒物初級來源和次級來源難以區分,排放量計算存在差異,分析方法不統一,這些還有待進一步提高改進。
4.2 已有大量關于雞舍和豬舍中顆粒物排放特點的研究。家畜舍類型、飼養方式、家畜類型、環境因子等均為影響顆粒物水平的重要因素。目前缺少標準化設備和方法來測量不同類型的畜禽舍顆粒物的濃度和排放水平。
4.3 畜禽舍中顆粒物吸附的刺激性氣體、惡臭化合物以及致病性和非致病性的微生物的種類需要進一步明確。
4.4 大量試驗研究了畜禽舍顆粒物對呼吸道的危害,但基于實驗條件限制,多為采集畜禽舍顆粒物在實驗室進行體外細胞試驗或以小鼠為模型的在體試驗,而缺少以畜禽為模型的在體試驗研究。
4.5 目前關于顆粒物對機體的損傷機制研究主要集中在顆粒物對呼吸道的致炎作用。但顆粒物成分復雜,并處在不斷變化中,因此顆粒物誘導機體損傷的機制也十分復雜,仍需進一步探究顆粒物對機體的損傷機理并總結出不同來源顆粒物對不同細胞損傷的共通性,以找出顆粒物引起細胞損傷的治療靶點和適合的緩解物質。
[1] AL HOMIDAN A, ROBERTSON J. Effect of litter type and stocking density on ammonia, dust concentrations and broiler performance., 2003, 44: 7-8.
[2] ANDERSEN C, VON ESSEN S G, SMITH L, SPENCER J, JOLIE R, DONHAM K J. Respiratory symptoms and airway obstruction in swine veterinarians: A persistent problem., 2004, 46(4): 386-392.
[3] SABINO R, FAíSCA V M, CAROLINO E, VERISSIMO C, VIEGAS C. Occupational exposure to aspergillus by swine and poultry farm workers in portugal., 2012, 75(22-23): 1381-1391.
[4] GRIMM E. Control of pm emission from livestock farming installations in germany., 2007: 21-26.
[5] KLIMONT Z, WAGNER F, AMANN M, COFALA J, SCH?PP W, HEYES C, BERTOK I, ASMAN W. The role of agriculture in the european commission strategy to reduce air pollution., 2007, 308: 3-10.
[6] CHARDON W, VAN DER HOEK K. Berekeningsmethode voor de emissie vanfijn stof vanuit de landbouw. (Calculation method for emission of fine dust from agriculture). Alterra/RIVM, Wageningen, The Netherlands, 2002: 35.
[7] EMEP/CORINAIR Atmospheric Emission Inventory Guidebook. December 2007 Update, third ed. EEA, Copenhagen, Denmark.
[8] CAMBRALóPEZ M, AARNINK A J, ZHAO Y, CALVET S, TORRES A G. Airborne particulate matter from livestock production systems: A review of an air pollution problem., 2010, 158(1): 1-17.
[9] HETLAND R B, CASSEE F R, L?G M, REFSNES M, DYBING E, SCHWARZE P E. Cytokine release from alveolar macrophages exposed to ambient particulate matter: Heterogeneity in relation to size, city and season., 2005, 2(1): 1-15.
[10] 沈丹, 戴鵬遠, 吳勝, 唐倩, 李春梅. 冬季封閉式肉種雞舍空氣顆粒物, 氨氣和二氧化碳分布特點及 PM 2.5 理化特性分析. 畜牧獸醫學報, 49(6): 1178-1193.
SHEN D, DAI P Y, WU S, TANG Q, LI C M. Distribution of particles, ammonia and carbon dioxide as well as physicochemical property of pm2.5in an enclosed broiler breeder house in winter., 49(6): 1178-1193.(in Chinese)
[11] ZHANG Y.. CRC Press, 2004.
[12] KULKARNI P, BARON P A, WILLEKE K. Aerosol fundamentals. In: Baron, P.A., Willeke, K.(Eds.),(seconded). Wiley, New York, 2001: 45–57.
[13] 劉志榮. 談談大氣顆粒物的分類和命名.中國科技術語, 2013(2): 31-34.
LIU Z R. Discuss the classification and naming of atmospheric particulates., 2013 (2): 31-34. (in Chinese)
[14] DONHAM K J, SCALLON L J, POPENDORF W, TREUHAFT M W, ROBERTS R C. Characterization of dusts collected from swine confinement buildings., 47(7): 404-410.
[15] CAMBRA-LóPEZ M, TORRES A, AARNINK A, OGINK N. Source analysis of fine and coarse particulate matter from livestock houses., 2011, 45(3): 694-707.
[16] CURTIS S E, DRUMMOND J G, GRUNLOH D J, LYNCH P B, JENSEN A H. Relative and qualitative aspects of aerial bacteria and dust in swine houses., 1975, 41(5): 1512-1520.
[17] HONEY L, MCQUITTY J. Some physical factors affecting dust concentrations in a pig facility., 1979, 21(1): 9-14.
[18] AMADOR I R, PINTO J P, SOLCI M C. Concentration and size distribution of particulate matter in a broiler house ambient air., 2016, 8(3): 189-193.
[19] QI R, MANBECK H, MAGHIRANG R. Dust net generation rate in a poultry layer house., 1992, 35(5): 1639-1645.
[20] AARNINK A, STOCKHOFE-ZURWIEDEN N, WAGEMANS M. Dust in different housing systems for growing-finishing pigs., 2004.
[21] XUELAN L, ZHANG Y, PEIPEI Y, QINGCHUAN J, XIANGFA W, RUITING L, TIANHONG S, BIN W. Effects of different padding on air quality in broiler house and growth physiological index of broilers., 2015, 16(12):2764-2769.
[22] SEEDORF J, HARTUNG J. Emission of airborne particulates from animal production., 2000.
[23] CHAI L, ZHAO Y, XIN H, WANG T, ATILGAN A, SOUPIR M, LIU K. Reduction of particulate matter and ammonia by spraying acidic electrolyzed water onto litter of aviary hen houses–a lab-scale study.:, 2016.
[24] SHEPHERD T A, ZHAO Y, LI H, STINN J P, HAYES M D, XIN H. Environmental assessment of three egg production systems—part ii. Ammonia, greenhouse gas, and particulate matter emissions., 2015, 94(3): 534-543.
[25] SHEN D, WU S, DAI P Y, TANG Q, LI C M. Distribution of particulate matter and ammonia and physicochemical properties of fine particulate matter in a layer house., in press.
[26] HARTUNG J, SALEH M. Composition of dust and effects on animals.2007,308: 111-116.
[27] SCHNEIDER T, SCHLüNSSEN V, VINZENTS P, KILDES? J. Passive sampler used for simultaneous measurement of breathing zone size distribution, inhalable dust concentration and other size fractions involving large particles., 2002, 46(2): 187-195.
[28] XU W, ZHENG K, MENG L, LIU X, HARTUNG E, ROELCKE M, ZHANG F. Concentrations and emissions of particulate matter from intensive pig production at a large farm in north china., 2016, 16(1): 79-90.
[29] CAMBRA-LóPEZ M, TORRES A, AARNINK A, OGINK N. Source analysis of fine and coarse particulate matter from livestock houses., 2011, 45: 694-707.
[30] ELLEN H, BOTTCHER R, VON WACHENFELT E, TAKAI H. Dust levels and control methods in poultry houses., 2000, 6(4): 275-282.
[31] HAEUSSERMANN A, COSTA A, AERTS J-M, HARTUNG E, JUNGBLUTH T, GUARINO M, BERCKMANS D. Development of a dynamic model to predict pm emissions from swine houses., 2008, 37(2): 557-564.
[32] 吳勝, 沈丹, 唐倩, 戴鵬遠,李延森,李春梅. 規模化半封閉式豬場舍內顆粒物、氨氣和二氧化碳分布規律. 畜牧與獸醫, 2018(3): 30-38.
WU S, SHEN D, TANG Q, DAI P Y, LI Y S, LI C M. Distribution of particulate matters and noxious gases in large-scale and semi-enclosed swine houses., 2018, 50(3): 30-38. (in Chinese)
[33] BANHAZI T, SEEDORF J, LAFFRIQUE M, RUTLEY D. Identification of the risk factors for high airborne particle concentrations in broiler buildings using statistical modelling., 2008, 101(1): 100-110.
[34] PUMA M, MAGHIRANG R, HOSNI M, HAGEN L. Modeling of dust concentration distribution in a simulated swine room under non-isothermal conditions., 1999, 42(6): 1823-1832.
[35] PUMA M, MAGHIRANG R, HOSNI M, HAGEN L. Modeling dust concentration distribution in a swine house under isothermal conditions., 1999, 42(6): 1811-1822.
[36] WOOD D J, VAN HEYST B J. A review of ammonia and particulate matter control strategies for poultry housing., 2016, 59(1): 329-344.
[37] CAMBRA-LóPEZ M, AARNINK A J, ZHAO Y, CALVET S, TORRES A G. Airborne particulate matter from livestock production systems: A review of an air pollution problem., 2010, 158(1): 1-17.
[38] HINZ T, LINKE S, KARLOWSKI J, MYCZKO R, KUCZYNSKI T, BERK J. PM emissions in and from force-ventilated turkey and dairy cattle houses as factor of health and the environment., 2007: 306-405.
[39] HINZ T, LINKE S, BITTNER P, KARLOWSKI J, KOLODZIEJCZYK T. Measuring particle emissions in and from a polish cattle house., 2007.
[40] HINZ T, LINKE S. A comprehensive experimental study of aerial pollutants in and emissions from livestock buildings. Part 2: Results., 1998, 70(1): 119-129.
[41] YODER M, VAN WICKLEN G. Respirable aerosol generation by broiler chickens., 1988, 31(5): 1510-1517.
[42] TAKAI H, PEDERSEN S, JOHNSEN J O, METZ J, KOERKAMP P G, UENK G, PHILLIPS V, HOLDEN M, SNEATH R, SHORT J. Concentrations and emissions of airborne dust in livestock buildings in northern europe., 1998, 70(1): 59-77.
[43] WATHES C, HOLDEN M, SNEATH R, WHITE R, PHILLIPS V. Concentrations and emission rates of aerial ammonia, nitrous oxide, methane, carbon dioxide, dust and endotoxin in uk broiler and layer houses., 1997, 38(1): 14-28.
[44] REDWINE J S, LACEY R E, MUKHTAR S, CAREY J. Concentration and emissions of ammonia and particulate matter in tunnel–ventilated broiler houses under summer conditions in texas., 2002, 45(4): 1101-1109.
[45] Al HOMIDAN A, ROBERTSON J F, PETCHEY A M. Effect of environmental factors on ammonia and dust production and broiler performance., 1998, 39: S9-S10.
[46] ELLEN H H, DOLEGHS B, ZOONS J. Influence of air humidity on dust concentration in broiler houses., 1999, Aarhus, Denmark.
[47] VUCEMILO M, MATKOVIC K, VINKOVIC B, MACAN J, VARNAI V M, PRESTER L J, GRANIC K, ORCT T. Effect of microclimate on the airborne dust and endotoxin concentration in a broiler house., 2008, 53 (2): 83-89.
[48] GUSTAFSSON G, VON WACHENFELT E. Airborne dust control measures for floor housing system for laying hens., 2006, VII: 1-13.
[49] HEBER A J, STROIK M, NELSSEN J L, NICHOLS D A. Influence of environmental factors on concentrations and inorganic content of aerial dust in swine finishing buildings., 1998(b), 31 (3): 875-881.
[50] MAGHIRANG R G, PUMA M C, LIU Y, CLARK P. Dust concentrations and particle size distribution in an enclosed swine nursery., 1997, 40 (3): 749-754.
[51] GUSTAFSSON G. Factors affecting the release and concentration of dust in pig houses., 1999, 74 (4): 379-390.
[52] HAEUSSERMANN A, FISHER D, JUNGBLUTH T, BAUR J, HARTUNG E. Aerosol indoor concentration and particulate emission in fattening pig husbandry., 2006, AgEng Bonn, Germany, 2006.
[53] VIEGAS S, FAíSCA V M, DIAS H, CLéRIGO A, CAROLINO E, VIEGAS C. Occupational exposure to poultry dust and effects on the respiratory system in workers., 2013, 76(4-5): 230-239.
[54] FRANZI L M, LINDERHOLM A L, RABOWSKY M, LAST J A. Lung toxicity in mice of airborne particulate matter from a modern layer hen facility containing proposition 2-compliant animal caging., 2016,33(3):211-221.
[55] HARRY E. Air pollution in farm buildings and methods of control: A review., 1978, 7(4): 441-454.
[56] HOUSE J S, WYSS A B, HOPPIN J A, UMBACH D M, LONDON S. Early-life farming exposures and adult atopy: The agricultural lung health study.:, 2016: A6695.
[57] RAASCHOU-NIELSEN O, BEELEN R, WANG M, HOEK G, ANDERSEN Z, HOFFMANN B, STAFOGGIA M, SAMOLI E, WEINMAYR G, DIMAKOPOULOU K. Particulate matter air pollution components and risk for lung cancer., 2016, 87: 66-73.
[58] SKóRA J, MATUSIAK K, WOJEWóDZKI P, NOWAK A, SULYOK M, LIGOCKA A, OKRASA M, HERMANN J, GUTAROWSKA B. Evaluation of microbiological and chemical contaminants in poultry farms., 2016, 13(2): 1-16.
[59] LI Q-F, WANG-LI L, LIU Z, JAYANTY R, SHAH S B, BLOOMFIELD P. Major ionic compositions of fine particulate matter in an animal feeding operation facility and its vicinity., 2014, 64(11): 1279-1287.
[60] MOSTAFA E, NANNEN C, HENSELER J, DIEKMANN B, GATES R, BUESCHER W. Physical properties of particulate matter from animal houses—empirical studies to improve emission modelling., 2016, 23(12): 12253-12263.
[61] ZHAO Y, AARNINK A J, DE JONG M C, GROOT KOERKAMP P W. Airborne microorganisms from livestock production systems and their relation to dust., 2014, 44(10): 1071-1128.
[62] NI L, CHUANG C C, ZUO L. Fine particulate matter in acute exacerbation of copd., 2015, 6: 1-10.
[63] SEEDORF J, HARTUNG J, SCHR?DER M, LINKERT K, PHILLIPS V, HOLDEN M, SNEATH R, SHORT J, WHITE R, PEDERSEN S. Concentrations and emissions of airborne endotoxins and microorganisms in livestock buildings in northern europe., 1998, 70(1): 97-109.
[64] SCHULZE A, VAN STRIEN R, EHRENSTEIN V, SCHIERL R, KUCHENHOFF H, RADON K. Ambient endotoxin level in an area with intensive livestock production., 2006, 13(1): 87-91.
[65] ZUCKER B A, MüLLER W. Concentrations of airborne endotoxin in cow and calf stables., 1998, 29(1): 217-221.
[66] SPAAN S, WOUTE RS I M, OOSTING I, DOEKES G, HEEDERIK D. Exposure to inhalable dust and endotoxins in agricultural industries., 2006, 8(1): 63-72.
[67] SCHUIJS M J, WILLART M A, VERGOTE K, GRAS D, DESWARTE K, EGE M J, MADEIRA F B, BEYAERT R, VAN LOO G, BRACHER F. Farm dust and endotoxin protect against allergy through a20 induction in lung epithelial cells., 2015, 349(6252): 1106-1110.
[68] LONDON S, HOPPIN J A, WYSS A, HOUSE J S, HENNEBERGER P K, UMBACH D M, THORNE P S, CARNES M U. House dust endotoxin levels are associated with adult asthma in the agricultural lung health study.:2016: A2781.
[69] MULLER-SUUR C, LARSSON K, MALMBERG P, LARSSON P. Increased number of activated lymphocytes in human lung following swine dust inhalation., 1997, 10(2): 376-380.
[70] SCHORLEMMER H, EDWARDS J, DAVIES P, ALLISON A. Macrophage responses to mouldy hay dust, micropolyspora faeni and zymosan, activators of complement by the alternative pathway., 1977, 27(2): 198-207.
[71] BAUER C, KIELIAN T, WYATT T A, ROMBERGER D J, WEST W W, GLEASON A M, POOLE J A. Myeloid differentiation factor 88-dependent signaling is critical for acute organic dust-induced airway inflammation in mice., 2013, 48(6): 781-789.
[72] DUSAD A, THIELE G M, KLASSEN L W, WANG D, DURYEE M J, MIKULS T R, STAAB E B, WYATT T A, WEST W W, REYNOLDS S J. Vitamin d supplementation protects against bone loss following inhalant organic dust and lipopolysaccharide exposures in mice., 2015, 62(1): 46-59.
[73] GOLDEN G A, WYATT T A, ROMBERGER D J, REIFF D, MCCASKILL M, BAUER C, GLEASON A M, POOLE J A. Vitamin d treatment modulates organic dust-induced cellular and airway inflammatory consequences., 2013, 27(1): 77-86.
[74] POOLE J A, WYATT T A, OLDENBURG P J, ELLIOTT M K, WEST W W, SISSON J H, VON ESSEN S G, ROMBERGER D J. Intranasal organic dust exposure-induced airway adaptation response marked by persistent lung inflammation and pathology in mice., 2009, 296(6): 1085-1095.
[75] POOLE J A, THIELE G M, ALEXIS N E, BURRELL A M, PARKS C, ROMBERGER D J. Organic dust exposure alters monocyte-derived dendritic cell differentiation and maturation., 2009, 297(4): 767-776.
[76] EASTMAN C, SCHENKER M B, MITCHELL D C, TANCREDI D J, BENNETT D H, MITLOEHNER F M. Acute pulmonary function change associated with work on large dairies in california., 2013, 55(1): 74-79.
[77] FENG S, GAO D, LIAO F, ZHOU F, WANG X. The health effects of ambient PM2.5and potential mechanisms., 2016, 128: 67-74.
[78] DENG X, WEI R, FANG Z, DING W. PM2.5induces Nrf2-mediated defense mechanisms against oxidative stress by activating PI3K/AKT signaling pathway in human lung alveolar epithelial A549 cells., 2013, 29(3): 143-157.
[79] LIMóN-PACHECO J, GONSEBATT M E. The role of antioxidants and antioxidant-related enzymes in protective responses to environmentally induced oxidative stress., 2009, 674(1): 137-147.
[80] HARRIS G K, SHI X. Signaling by carcinogenic metals and metal- induced reactive oxygen species., 2003, 533(1): 183-200.
[81] HUANG J, LAM G Y, BRUMELL J H. Autophagy signaling through reactive oxygen species., 2011, 14(11): 2215-2231.
[82] NAIK E, DIXIT V M. Mitochondrial reactive oxygen species drive proinflammatory cytokine production., 2011, 208(3): 417-420.
[83] DAVIS R J. Signal transduction by the JNK group of map kinases., 2000, 103(2): 239-252.
[84] MIYATA R, VAN EEDEN S F. The innate and adaptive immune response induced by alveolar macrophages exposed to ambient particulate matter., 2011, 257(2): 209-226.
[85] MARANO F, BOLAND S, BONVALLOT V, BAULIG A, BAEZA-SQUIBAN A. Human airway epithelial cells in culture for studying the molecular mechanisms of the inflammatory response triggered by diesel exhaust particles., 2002, 18(5): 315-320.
[86] GUNTER T E, GUNTER K K, SHEU S-S, GAVIN C E. Mitochondrial calcium transport: Physiological and pathological relevance., 1994, 267(2): 313-339.
[87] CHAN S-L, LINDQUIST L D, HANSEN M J, GIRTMAN M A, PEASE L R, BRAM R J. Calcium-modulating cyclophilin ligand is essential for the survival of activated t cells and for adaptive immunity., 2015, 195(12): 5648-5656.
[88] XING Y F, XU Y H, SHI M H, LIAN Y X. The impact of PM2.5on the human respiratory system., 2016, 8(1): 69-74.
[89] HE M, ICHINOSE T, YOSHIDA Y, ARASHIDANI K, YOSHIDA S, TAKANO H, SUN G, SHIBAMOTO T. Urban PM2.5exacerbates allergic inflammation in the murine lung via a TLR2/TLR4/ Myd88-signaling pathway., 2017, 7(1): 1-9.
[90] HE M, ICHINOSE T, REN Y, SONG Y, YOSHIDA Y, ARASHIDANI K, YOSHIDA S, NISHIKAWA M, TAKANO H, SUN G. Pm2.5-rich dust collected from the air in fukuoka, kyushu, japan, can exacerbate murine lung eosinophilia., 2015, 27(6): 287-299.
[91] MAHLA R S, REDDY C M, PRASAD D, KUMAR H. Sweeten pamps: Role of sugar complexed pamps in innate immunity and vaccine biology., 2013, 4: 1-16.
[92] JIMéNEZ-DALMARONI M J, GERSWHIN M E, ADAMOPOULOS I E. The critical role of toll-like receptors—from microbial recognition to autoimmunity: A comprehensive review., 2016, 15(1): 1-8.
[93] BECKER S, DAILEY L, SOUKUP J M, SILBAJORIS R, DEVLIN R B. Tlr-2 is involved in airway epithelial cell response to air pollution particles., 2005, 203(1): 45-52.
[94] FUERTES E, BRAUER M, MACINTYRE E, BAUER M, BELLANDER T, VON BERG A, BERDEL D, BRUNEKREEF B, CHAN-YEUNG M, GEHRING U. Childhood allergic rhinitis, traffic-related air pollution, and variability in the GSTP1 , TNF , TLR2 , and TLR4 genes: results from the TAG study., 2013, 132(2): 342-352.
[95] ZHAO C, LIAO J, CHU W, WANG S, YANG T, TAO Y, WANG G. Involvement of TLR2 and TLR4 and Th1/Th2 shift in inflammatory responses induced by fine ambient particulate matter in mice., 2012, 24(13): 918-927.
[96] SHOENFELT J, MITKUS R J, ZEISLER R, SPATZ R O, POWELL J, FENTON M J, SQUIBB K A, MEDVEDEV A E. Involvement of tlr2 and tlr4 in inflammatory immune responses induced by fine and coarse ambient air particulate matter., 2009, 86(2): 303-312.
[97] DEGUINE J, BARTON G M. Myd88: A central player in innate immune signaling., 2014, 6(97): 1-7.
[98] POOLE J A, WYATT T A, KIELIAN T, OLDENBURG P, GLEASON A M, BAUER A, GOLDEN G, WEST W W, SISSON J H, ROMBERGER D J. Toll-like receptor 2 regulates organic dust- induced airway inflammation., 2011, 45(4): 711-719.
[99] ZENG Y, YANG X, WANG J, FAN J, KONG Q, YU X. Aristolochic acid I induced autophagy extenuates cell apoptosis via ERK1/2 pathway in renal tubular epithelial cells., 2012, 7(1): 1-10.
[100] POON A H, CHOUIALI F, TSE S M, LITONJUA A A, HUSSAIN S N, BAGLOLE C J, EIDELMAN D H, OLIVENSTEIN R, MARTIN J G, WEISS S T. Genetic and histological evidence for autophagy in asthma pathogenesis., 2012, 129(2): 1-7.
[101] DENG X, ZHANG F, WANG L, RUI W, LONG F, ZHAO Y, CHEN D, DING W. Airborne fine particulate matter induces multiple cell death pathways in human lung epithelial cells., 2014, 19(7): 1099-1112.
[102] LIU T, WU B, WANG Y, HE H, LIN Z, TAN J, YANG L, KAMP D W, ZHOU X, TANG J. Particulate matter 2.5 induces autophagy via inhibition of the phosphatidylinositol 3-kinase/akt/mammalian target of rapamycin kinase signaling pathway in human bronchial epithelial cells., 2015, 12(2): 1914-1922.
[103] SU R, JIN X, ZHANG W, LI Z, LIU X, REN J. Particulate matter exposure induces the autophagy of macrophages via oxidative stress-mediated PI3K/AKT/mTOR pathway., 2017, 167: 444-453.
[104] WANG Y, LIN Z, HUANG H, HE H, YANG L, CHEN T, YANG T, REN N, JIANG Y, XU W. AMPK is required for PM2.5-induced autophagy in human lung epithelial A549 cells., 2015, 8(1): 1-15.
[105] JONES S A, MILLS K H, HARRIS J. Autophagy and inflammatory diseases., 2013, 91(3): 250-258.
[106] TAL M C, SASAI M, LEE H K, YORDY B, SHADEL G S, IWASAKI A. Absence of autophagy results in reactive oxygen species-dependent amplification of rlr signaling., 2009, 106(8): 2770-2775.
[107] FAN X, WANG J, HOU J, LIN C, BENSOUSSAN A, CHANG D, LIU J, WANG B. Berberine alleviates ox-LDL induced inflammatory factors by up-regulation of autophagy via AMPK/mTOR signaling pathway., 2015, 13(1): 92-103.
[108] DENG X, RUI W, ZHANG F, DING W. PM2.5induces Nrf2-mediated defense mechanisms against oxidative stress by activating PI3K/AKT signaling pathway in human lung alveolar epithelial A549 cells., 2013, 29(3): 143-157.
[109] 何賢輝, 何健, 歐陽東云. 細胞自噬與炎癥反應相互作用的研究進展.暨南大學學報: 自然科學與醫學版, 2013, 34(2): 125-128.
HE X H, HE J, OUYANG D Y. Research progress in the interaction between autophagy and inflammatory response.:, 2013, 34(2): 125-128. (in Chinese)
(責任編輯 林鑒非)
Research Progress on Characteristics of Particulate Matter in Livestock Houses and Its Harmful Effects on Respiratory Tract Health of Livestock and Poultry
DAI PengYuan, SHEN Dan, TANG Qian, LI YanSen, LI ChunMei
(College of Animal Science & Technology Nanjing Agricultural University,Nanjing 210095)
With the improvement of livestock and poultry intensive breeding, high density breeding livestock and poultry farms air quality problem becomes increasingly prominent. The livestock production process can generate a large number of PM, which has become an important source of fine particulate PM2.5(aerodynamic diameter ≤ 2.5 μm) and PM10(aerodynamic diameter ≤ 10 μm ) in atmosphere, affecting the air quality and the health of livestock seriously. PM in livestock houses is mainly organic and mainly from feed, feces, feathers, dander, containing C, H, O, N, S, Ca, Na, Mg, Al, K, and other elements; the surface of PM also adheres to bacteria, fungi, viruses, endotoxins, ammonia gas, hydrogen sulfide and other harmful substances. It was found that PM concentration in chicken house was higher than that in pig house; PM concentration in livestock houses was positively correlated with the age and activity of animals; PM concentration in winter was higher than that in summer. However, there is a lack of standardized equipment and standard methods to measure PM concentration and emission levels in different types of livestock and poultry houses. PM components in livestock houses are complex and have strong biological effects, which seriously hazard the health and animal production. High PM concentration in livestock houses affects respiratory health mainly in the following three forms: the directly stimulation of PM to respiratory tract which reduces the immune resistance of the body to respiratory diseases; the stimulation of various compounds attached to PM surface; the stimulation of pathogenic and non-pathogenic microorganisms on PM surfaces. At present, studies on the mechanism of PM on respiratory health hazards mainly focus on the inflammatory effect of PM on respiratory tract Studies showed that, PM could induce cells to release inflammatory factors and cause lung inflammation reaction through the proinflammatory factor produced by alveolar macrophages stimulated by PM. In addition, PM2.5upregulated the expression of nuclear transcription factor κB (NFκB) and transcription activator AP-1 by mitogen-activated protein kinase (MAPKs) which was activated by oxidative stress. PM2.5could also identify the toll-like receptor 2 and 4 by activating the mode of activation, and activate the NFκB signaling pathway, leading to the occurrence of inflammation. Studies have also found that the cell autophagy and nuclear factor related factor - 2 (nuclear factor E2 - related factor 2, Nrf2) related signaling pathways would be activated during the process of inflammation induced by PM2.5, which provided the targets for treatment of cell damage induced by PM2.5. Although there were more study on mechanism of hazard of PM to the health of respiratory tract, the PM composition was complicated, and in a constantly changing, so the PM induced respiratory damage mechanism was very complex and need further study. A large number of PM released in the process of livestock production seriously affects the environmental air quality and the health of livestock, and the extent of PM's harm to the environment and the health of livestock was closely related to its composition and concentration. Therefore, a proper understanding of PM morphology, size, composition, concentration level and emission influencing factors of animal house is of great significance to the determination of PM source and hazard caused by PM toxicity. In this paper, the source, chemical composition, concentration, discharge, influence factors, and the effects on respiratory function of PM from animal house are summarized, and offer a base for evaluating the effect of PM on healthy production of livestock and poultry.
livestock house; particulate matter; pollution characteristics; respiratory tract damage
2018-04-11;
2018-07-17
“十三五”國家重點研發計劃(2016YFD0500505);國家自然科學基金(31772648)
戴鵬遠,Tel:025-84395971;E-mail:2015205015@njau.edu.cn。
李春梅,Tel:025-84395971;E-mail:chunmeili@njau.edu.cn
10.3864/j.issn.0578-1752.2018.16.017
