外源硫誘導(dǎo)下的Desulfovibrio desulfuricans sub sp. EPS特性及對(duì)Zn(Ⅱ)的吸附
甘 雨,宋衛(wèi)鋒,楊佐毅,連澤陽(yáng),馬雙念,黃祥武,羊仁高,溫炎標(biāo)
外源硫誘導(dǎo)下的sp. EPS特性及對(duì)Zn(Ⅱ)的吸附
甘 雨,宋衛(wèi)鋒*,楊佐毅,連澤陽(yáng),馬雙念,黃祥武,羊仁高,溫炎標(biāo)
(廣東工業(yè)大學(xué)環(huán)境科學(xué)與工程學(xué)院,廣東 廣州 510006)
研究了3種外源硫(Na2SO4、Na2SO3和Na2S2O3·5H2O)對(duì)sp.(.sp.)的胞外聚合物(EPS)的脅迫/誘導(dǎo)作用.結(jié)果表明,在還原性硫源0.50g/L Na2SO3的條件下,EPS產(chǎn)量最高,為2104.39mg/g VSS,蛋白質(zhì)含量為1888.52mg/g VSS,較脅迫/誘導(dǎo)前均提高了300%以上;其對(duì)Zn(Ⅱ)的吸附性能最好,為954.4mg/g EPS,提高了98.17%.三維熒光(3D-EEM)結(jié)果表明,脅迫/誘導(dǎo)后EPS中類(lèi)酪氨酸均大量增加;傅里葉紅外光譜(FTIR)結(jié)果表明,脅迫后-OH、C=O、C-O-C等官能團(tuán)均大量增加,在Zn(Ⅱ)的吸附中發(fā)揮了重要作用;X光電子能譜(XPS)結(jié)果表明,在還原性硫源(Na2SO3和Na2S2O3·5H2O)脅迫/誘導(dǎo)后,EPS中C-O/C-N、C=N和某種含氧基團(tuán)(X)大量增加,可能是吸附Zn(Ⅱ)的主要基團(tuán).
外源硫;脅迫/誘導(dǎo);EPS;Zn(Ⅱ)
胞外聚合物(EPS)存在于細(xì)胞外和微生物聚集體內(nèi)部,為細(xì)胞聚集以至形成顆粒污泥的關(guān)鍵,主要由蛋白質(zhì)、多糖、腐殖質(zhì)、核酸、脂質(zhì)和磷脂組成,其中蛋白質(zhì)和多糖是主要成分[1-2].產(chǎn)生EPS是細(xì)菌的一種自我保護(hù)機(jī)制,有利于細(xì)菌在不良條件中生存[3],其中含有豐富的官能團(tuán),如羧基(-COOH)、氨基(-NH2)、羥基(-OH)和羰基(C=O)等,可以和重金屬離子結(jié)合,防止重金屬離子進(jìn)入細(xì)胞內(nèi)[4].EPS的產(chǎn)生受多種因素影響,如營(yíng)養(yǎng)元素碳、氮、磷,以及生長(zhǎng)環(huán)境如pH值、溫度等[5].近年來(lái),關(guān)于EPS脅迫/誘導(dǎo)研究主要集中在重金屬方面[6-8],脅迫后EPS中官能團(tuán)的濃度與重金屬的濃度具有相關(guān)性,如Zn(Ⅱ)與-OH、C-O-C等官能團(tuán)[9],還發(fā)現(xiàn)類(lèi)色氨酸參與了Cr(Ⅵ)的還原[10].重金屬脅迫/誘導(dǎo)不僅能提高EPS產(chǎn)量還可以降低其他重金屬的毒性[11],SRB在Zn(Ⅱ)脅迫/誘導(dǎo)后,EPS對(duì)Zn(Ⅱ)、Cu(Ⅱ)和Cd(Ⅱ)的吸附量都增加了[12].但由于重金屬的毒性,難以得到大量的EPS.研究者逐漸將視野轉(zhuǎn)向營(yíng)養(yǎng)物質(zhì),如碳源[13]和氮源[14].一些特殊的物質(zhì)還可以定向增加官能團(tuán)的含量,在外源硫Na2S的脅迫/誘導(dǎo)下EPS中蛋白質(zhì)增加了將近一倍,尤其是蛋白質(zhì)中的巰基(-SH) 增加了約48.2%,EPS重金屬的吸附能力也變得更加突出[15].不僅如此,氧化/還原性物質(zhì)也可以起到一定的脅迫/誘導(dǎo)作用[16].因此,EPS的脅迫/誘導(dǎo)因子多種多樣,不應(yīng)局限于重金屬.
目前,國(guó)內(nèi)外對(duì)EPS的研究主要集中在好氧菌EPS的脅迫/誘導(dǎo)效應(yīng)上[17-18],對(duì)厭氧菌EPS的脅迫/誘導(dǎo)效應(yīng)研究較少.硫酸鹽還原菌(SRB)屬于革蘭氏陰性厭氧菌,以乳酸或丙酮酸等碳源為電子供體將硫酸鹽、亞硫酸鹽、硫代硫酸鹽等物質(zhì)還原為硫化氫[19],釋放出的硫離子與吸附在EPS上的重金屬離子形成金屬硫化物沉淀.含硫無(wú)機(jī)鹽作為SRB的營(yíng)養(yǎng)物質(zhì),為SRB的生命活動(dòng)提供能量,如亞硫酸鹽、硫代硫酸鹽可發(fā)生歧化反應(yīng)生成硫酸鹽并釋放能量[20],也必將促進(jìn)EPS的合成.硫源對(duì)于SRB產(chǎn)生EPS有重要意義,但相應(yīng)的的研究報(bào)道幾乎沒(méi)有.
研究EPS與重金屬作用的意義不僅體現(xiàn)在重金屬的去除,也體現(xiàn)在生物合成金屬硫化物中[21],其中Zn(Ⅱ)具有重要價(jià)值,如SRB胞外聚合物中的多糖和蛋白質(zhì)均可以提供Zn(Ⅱ)的結(jié)合點(diǎn)位,可以更高效的進(jìn)行吸附,而Cu(Ⅱ)只能與多糖結(jié)合[22].不僅如此,EPS與Zn(Ⅱ)的作用是生物合成具有特殊性能的ZnS量子點(diǎn)的重要一環(huán)[21].在以往研究中,使用EDTA-4Na+降低Zn(Ⅱ)的毒性[23],但是EDTA也會(huì)與EPS發(fā)生螯合,一定程度上阻礙了生物合成.脅迫/誘導(dǎo)可能會(huì)提高SRB對(duì)Zn(Ⅱ)的吸附能力,也就可能通過(guò)脅迫/誘導(dǎo)促進(jìn)生物合成ZnS.
本研究所使用菌種為脫硫弧菌脫硫亞種(sp.),以3種外源硫(Na2SO4、Na2SO3、Na2S2O3·5H2O)為脅迫/誘導(dǎo)因子,在同一濃度梯度下對(duì).sp.進(jìn)行培養(yǎng),探究其生長(zhǎng)情況、EPS產(chǎn)量及組分和吸附性能的變化,并通過(guò)多種測(cè)試分析方法揭示脅迫/誘導(dǎo)作用規(guī)律.研究發(fā)現(xiàn)EPS的產(chǎn)量大量增加,且作為主要成分的蛋白質(zhì)產(chǎn)量極高,對(duì)高效處理重金屬?gòu)U水有啟發(fā)作用.
1 材料與方法
1.1 材料
菌種為.sp.,由北京百歐博偉生物科技有限公司提供,經(jīng)平板劃線(xiàn)法厭氧培養(yǎng)3~4代后接種至液體培養(yǎng)基厭氧活化培養(yǎng),然后用甘油于-80.00℃中保存待用.
厭氧血瓊脂培養(yǎng)基:從廣州翔博生物科技有效公司購(gòu)得,由一次性無(wú)菌塑料平皿和瓊脂培養(yǎng)基組成.其中,每1.00L瓊脂含10.00g酪蛋白胰酶消化物、400.00mL半胱氨酸、1.00g玉米淀粉、14.00g瓊脂、0.01g維生素K1、5.00g酵母浸出粉,5.00g氯化鈉、70.00mL羊血或馬血,加蒸餾水至1.00L配制而成.
Starkey培養(yǎng)基[24]:稱(chēng)取0.50g K2HPO4、1.00g NH4Cl、1.00g Na2SO4、0.10g CaCl2·2H2O、2.00g MgSO4·7H2O 、2.00g DL-乳酸鈉、1.00g酵母粉,用超純水定容至1.00L,調(diào)節(jié)pH值為(7.00±0.20),于錐形瓶中121℃高壓滅菌20min,冷卻至常溫.使用前加入用無(wú)菌水配置的抗壞血酸,使其在培養(yǎng)基中的濃度為0.10g/L.初始Starkey培養(yǎng)基中不含SO32-和S2O32-,初始SO42-濃度為1.46g/L.
外源硫溶液:誘導(dǎo)/脅迫所使用的外源硫?yàn)镾O42-、SO32-和S2O32-,以Na2SO4、Na2SO3和Na2S2O3·5H2O溶液的形式加入到培養(yǎng)基中.
1.2 活化與培養(yǎng)
將.sp.凍干粉用0.20mL無(wú)菌水溶解,用平板劃線(xiàn)法接種至厭氧血瓊脂培養(yǎng)基中,裝入?yún)捬醮⒊掷m(xù)吹入氮?dú)?min,于30℃恒溫培養(yǎng)箱中厭氧活化培養(yǎng)48h,再挑取單個(gè)菌落重復(fù)上述厭氧活化培養(yǎng)步驟3~4次,確保菌種復(fù)蘇,恢復(fù)活性.
然后挑取單個(gè)菌落接種至Starkey液體培養(yǎng)基中,持續(xù)吹入氮?dú)?min后密封,于35℃,150r/min的條件下恒溫震蕩培養(yǎng)48h后,用甘油于-80℃(Haier 醫(yī)用低溫保存箱DW-86L338J)中保存待用.
將菌種解凍至常溫后以5.00%()接種至Starkey液體培養(yǎng)基中,持續(xù)吹入氮?dú)?min后密封,于35℃,150r/min的條件下恒溫震蕩培養(yǎng)24h后,再以10.00%()分別接種到濃度為0,0.10,0.20,0.30, 0.40,0.50,0.60,0.70,0.80,0.90g/L不同外源硫的Starkey液體培養(yǎng)基中,持續(xù)吹入氮?dú)?min后密封,于35℃,150r/min的條件下恒溫震蕩厭氧培養(yǎng)72h,達(dá)到穩(wěn)定期[24].上述接種過(guò)程均在無(wú)菌條件下進(jìn)行.
1.3 EPS提取與主要成分測(cè)定
采用NaOH法提取EPS[24].取30.00mL經(jīng)外源硫脅迫/誘導(dǎo)培養(yǎng)后的菌液,在8000r/min,4℃的條件下離心10min;棄掉上清液,加入30.00mL的0.90%NaCl,震蕩洗滌菌體,再于上述相同條件下離心10min;棄掉上清液,加入20.00mL的0.90%NaCl,震蕩洗滌菌體,再以5.00%()加入1.00ml的1.00mol/L NaOH,搖勻,于4℃條件下靜止3h;于4℃下,分別在16000r/min和8000r/min的條件下,各離心20min和15min;取上清液于0.45μm過(guò)濾器中過(guò)濾,再置于4000Da透析袋中透析24h,提純EPS后于-20℃下保存?zhèn)溆?
EPS產(chǎn)量用蛋白質(zhì)、多糖、核酸三者之和表示,3種成分分別用考馬斯亮藍(lán)法、硫酸蒽酮法、二苯胺法進(jìn)行測(cè)定.EPS產(chǎn)量的實(shí)驗(yàn)結(jié)果取3次平行實(shí)驗(yàn)的平均值.
1.4 EPS表征分析
1.4.1 三維熒光光譜(3D-EEM)分析 用Edinburgh FLS1000型熒光分光光度計(jì)測(cè)定,對(duì)EPS主要成分進(jìn)行比較分析,激發(fā)光(x)和發(fā)射光(m)掃描范圍分別為200~450nm、200~550nm.
1.4.2 X射線(xiàn)光電子能譜(XPS)分析 采用K- Alpha型X射線(xiàn)光電子能譜儀(賽默飛,英國(guó))對(duì)EPS進(jìn)行測(cè)試分析.能譜掃描范圍為0.00~1200.00eV,能譜采用C 1s(284.80eV校正),分辨率:pass energy 100.00eV,C 1s和O 1s等分辨率為40.00eV.所有的峰都用C 1s峰的結(jié)合能在284.80eV校準(zhǔn).
1.4.3 傅里葉紅外光譜(FTIR)分析 采用Thermo Scientific Nicolet iS20型傅里葉變換紅外光譜儀對(duì)EPS進(jìn)行測(cè)試分析.掃描范圍為400~4000cm-1,分辨率為4cm-1,樣品掃描次數(shù)為32.
1.5 Zn(Ⅱ)吸附實(shí)驗(yàn)
配制濃度為20.00mg/L的Zn(Ⅱ)溶液,調(diào)節(jié)pH值為5.00,取不同脅迫/誘導(dǎo)條件得到的等質(zhì)量(0.10mg)的EPS于錐形瓶中,并加入上述Zn(Ⅱ)溶液15.00mL.于35℃、150r/min條件下震蕩吸附2h,然后將混合物裝入分子量為4000Da的透析袋中,于200.00mL的超純水中透析12h.用火焰原子吸收分光光度計(jì)測(cè)定透析后樣品中Zn(Ⅱ)的濃度.吸附實(shí)驗(yàn)的結(jié)果均取3次實(shí)驗(yàn)結(jié)果的平均值.EPS吸附量的公式如下:
= (00-CV)/(1)
式中:為重金屬吸附量,mg/g EPS;0為金屬離子初始濃度,mg/L;0為初始溶液體積,L;C為吸附后金屬離子濃度,mg/L;V為透析液體積,L;為EPS質(zhì)量,g.
2 結(jié)果與討論
采用3種不同的外源硫作為脅迫/誘導(dǎo)因子,在0~0.90g/L的濃度梯度下對(duì).sp.進(jìn)行脅迫/誘導(dǎo),相應(yīng)的EPS分別記為Na2SO4-EPS,Na2SO3-EPS,Na2S2O3-EPS,空白樣記為Control-EPS.
2.1 脅迫/誘導(dǎo)下D. desulfuricans sp.的生長(zhǎng)情況
為了方便對(duì)比,本文中以外源硫的濃度來(lái)表示脅迫/誘導(dǎo)強(qiáng)度,由圖1可看出,.sp在3種不同外源硫的脅迫/誘導(dǎo)下,總體趨勢(shì)是細(xì)胞干重隨著脅迫/誘導(dǎo)濃度的增加先減少后增加.其中,Na2SO3對(duì)于細(xì)菌的生長(zhǎng)影響最大,在Na2SO3脅迫/誘導(dǎo)下細(xì)胞干重基本上低于相同濃度的Na2SO4和Na2S2O3·5H2O,當(dāng)Na2SO3濃度達(dá)到0.50g/L時(shí),細(xì)胞干重為0.40g/L,相較于脅迫/誘導(dǎo)前,降低了44.51%.當(dāng)脅迫/誘導(dǎo)濃度達(dá)到1.20g/L時(shí),在Na2SO4和Na2S2O3·5H2O條件下,細(xì)菌大量繁殖,細(xì)胞干重大幅增加.Na2SO4脅迫/誘導(dǎo)下細(xì)胞干重最高,達(dá)到1.42g/L,增幅為106.37%.Na2SO3的條件下,細(xì)胞干重增長(zhǎng)趨勢(shì)十分緩慢.
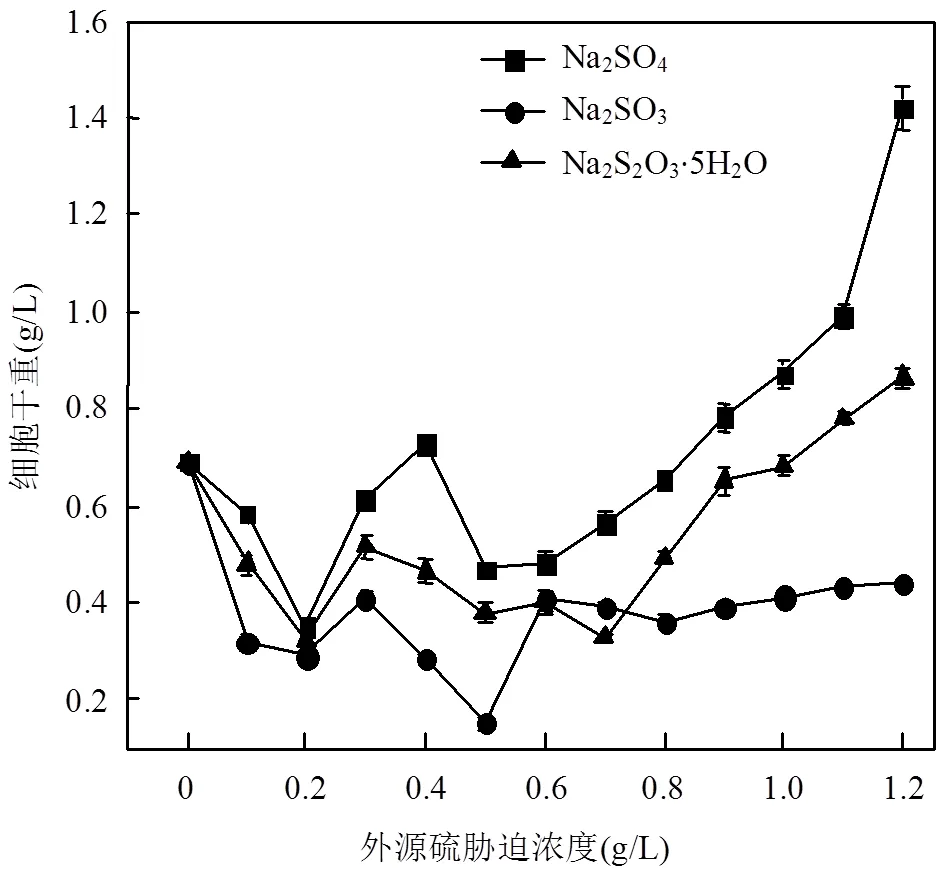
圖1 不同濃度外源硫脅迫/誘導(dǎo)下D. desulfuricans sp.單位體積培養(yǎng)基的細(xì)胞干重
2.2 脅迫/誘導(dǎo)下D. desulfuricans sp.EPS組分變化
由圖2可知,雖然在Na2SO3脅迫/誘導(dǎo)下.sp.細(xì)胞干重減少了,但在0.50g/L時(shí),其單位質(zhì)量微生物的EPS產(chǎn)量卻是最高的,EPS的產(chǎn)量從脅迫/誘導(dǎo)前的494.38mg/g VSS提高到2104.39mg/g VSS,提高了325.66%,其中蛋白質(zhì)的產(chǎn)量從451.44mg/g VSS提高到了1888.52mg/g VSS,提高了318.33%,整體上單位體積培養(yǎng)基的EPS產(chǎn)量相差并不明顯.
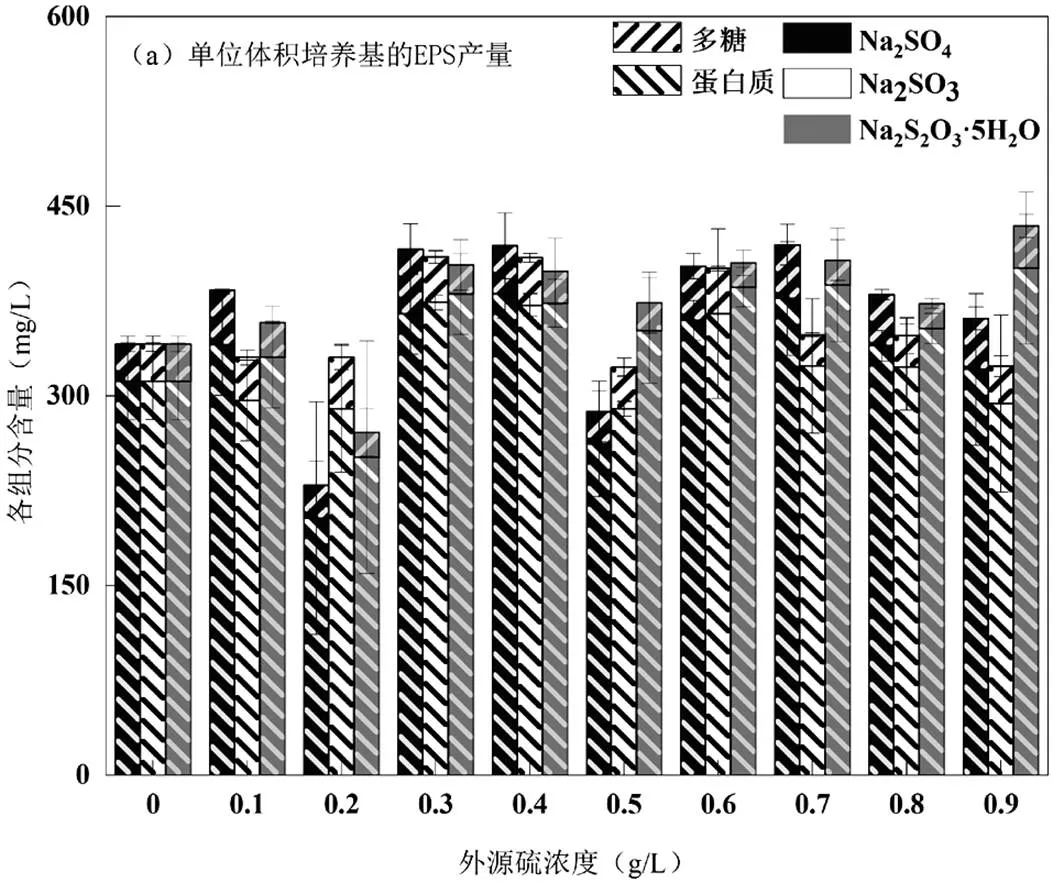
Na2SO4為0.60g/L時(shí),EPS產(chǎn)量從494.38mg/g VSS提高到833.13mg/g,提高了68.52%,其中蛋白質(zhì)從451.44mg/g VSS提高到744.28mg/g VSS,提高了74.71%;Na2S2O3·5H2O為0.70g/L時(shí),EPS的產(chǎn)量從脅迫/誘導(dǎo)前的494.38mg/g VSS提高到1247.39mg/g VSS,提高了152.31%,其中蛋白質(zhì)產(chǎn)量從451.44mg/g VSS提高到了1186.94mg/g VSS,提高了162.92%.另外,3種條件下EPS中的多糖變化不明顯,但其產(chǎn)量同樣在相應(yīng)的最佳脅迫/誘導(dǎo)濃度下達(dá)到最大(Na2SO4:88.86mg/g VSS、Na2SO3:215.87mg/g VSS、Na2S2O3·5H2O:60.46mg/g VSS),而DNA未檢測(cè)出.
由上述可知,在3種外源硫中,還原性硫源Na2SO3對(duì).sp.的脅迫/誘導(dǎo)最為突出, Na2S2O3·5H2O次之,Na2SO4較差.Na2SO3超過(guò)最佳脅迫/誘導(dǎo)濃度,EPS和蛋白質(zhì)的產(chǎn)量會(huì)迅速降低至最高值的46.81%、47.51%,隨著濃度增加會(huì)變得更低.由此可見(jiàn)Na2SO3既能顯著提高.sp.EPS產(chǎn)量,超過(guò)一定范圍也能顯著抑制EPS的產(chǎn) 生.
在SRB還原硫酸鹽途徑中,SRB需要消耗能量還原硫酸鹽,SRB可以將亞硫酸鹽或者硫代硫酸鹽歧化,生成硫酸鹽,同時(shí)釋放能量供其生命活動(dòng)所需.在本研究中,Na2SO3脅迫效果最好極有可能是其能夠提供更多的能量以合成氨基酸,而硫代硫酸鹽歧化釋放的能量較少,所以脅迫效果次之.已有研究表明,亞硫酸鹽歧化釋放的能量遠(yuǎn)大于硫代硫酸鹽[20].
2.3 脅迫/誘導(dǎo)下D. desulfuricans sp. EPS的吸附性能
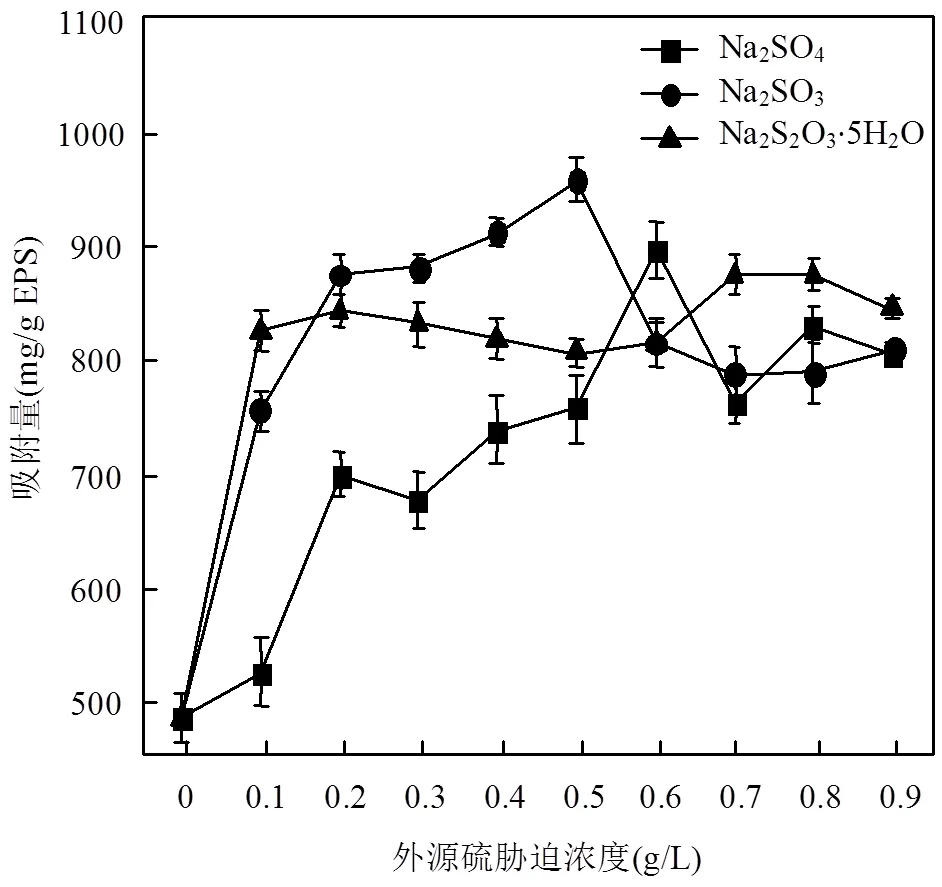
圖3 不同濃度外源硫脅迫下EPS對(duì)Zn(Ⅱ)的吸附量
由圖3可知,脅迫/誘導(dǎo)下的EPS對(duì)Zn(Ⅱ)的吸附量在整體上均體現(xiàn)出先增加后減少的趨勢(shì). Na2SO4最佳脅迫/誘導(dǎo)濃度為0.60g/L時(shí),吸附量從脅迫/誘導(dǎo)前的481.6mg/g EPS增加到了893.6mg/g EPS,提高了85.54%;Na2SO3為0.50g/L時(shí),吸附量增加到954.4mg/g EPS,提高了98.17%;Na2S2O3·5H2O為0.70g/L時(shí),吸附量增加到871.8mg/g EPS,提高了81.02%.
Na2SO3-EPS對(duì)Zn(Ⅱ)的吸附能力較強(qiáng), Na2SO4-EPS的吸附能力次之,Na2S2O3-EPS的吸附能力較差.結(jié)合EPS產(chǎn)量和組分變化規(guī)律,Na2SO3對(duì).sp.的脅迫/誘導(dǎo)效果最為明顯,不僅大幅提高了EPS產(chǎn)量(尤其是蛋白質(zhì)),且對(duì)Zn(Ⅱ)的吸附能力也成倍增加.當(dāng)脅迫/誘導(dǎo)濃度超過(guò)一定范圍,不僅EPS產(chǎn)量減少,且吸附Zn(Ⅱ)能力也減弱.
總體上看,Na2SO3、Na2SO4和Na2S2O3·5H2O脅迫/誘導(dǎo)可以提高EPS的重金屬吸附性能,且吸附性能變化趨勢(shì)與組分變化規(guī)律基本一致.
2.4 脅迫/誘導(dǎo)前后EPS三維熒光分析
用三維熒光光譜研究3種外源硫的最佳脅迫/誘導(dǎo)前后的EPS.由于EPS樣品中含水,測(cè)試結(jié)果均減去空白水樣的背景值.
由圖4和表1可知,Peak A強(qiáng)度較高而Peak B強(qiáng)度較低,它們分別為可溶性微生物產(chǎn)物(SMP)和酪氨酸,均含有C=O、-NH2/NH、-COOH、-OH等官能團(tuán),可以與重金屬離子進(jìn)行螯合和吸附.
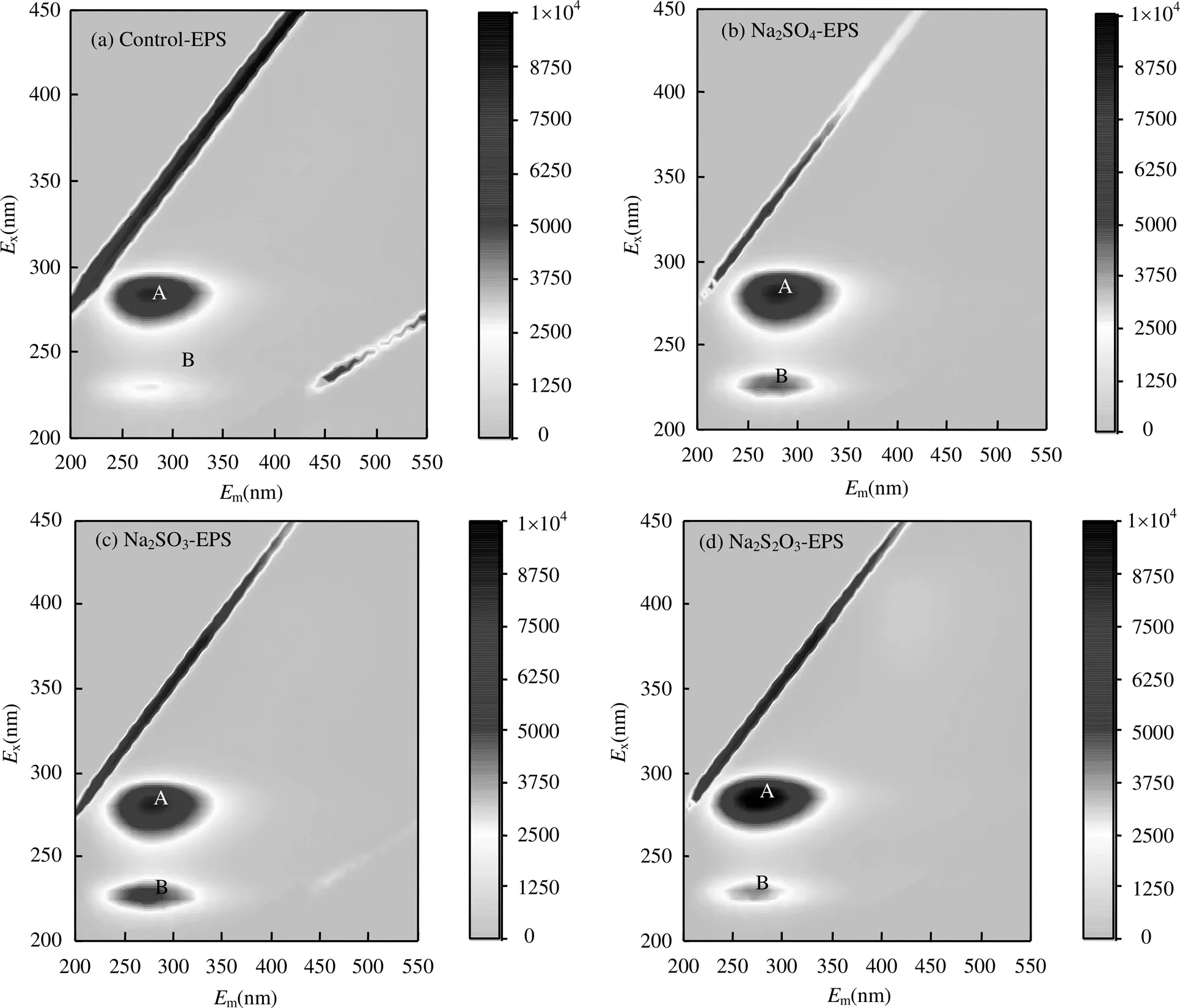
圖4 外源硫脅迫/誘導(dǎo)前后EPS三維熒光譜圖
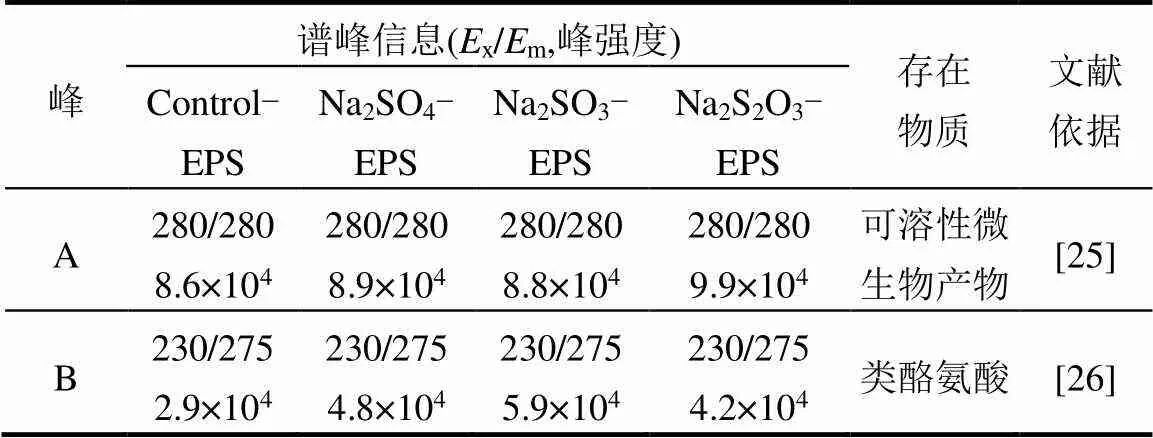
表1 熒光光譜中的譜峰信息及其對(duì)應(yīng)成分
Na2SO4的脅迫/誘導(dǎo)下,反映蛋白類(lèi)物質(zhì)的Peak B強(qiáng)度與Control-EPS相比,提高65.51%; Na2SO3的脅迫/誘導(dǎo)下,Peak B強(qiáng)度提高103.45%; Na2S2O3·5H2O脅迫/誘導(dǎo)下,Peak B強(qiáng)度提高44.83%,而Peak A強(qiáng)度增長(zhǎng)緩慢.由此推斷,類(lèi)酪氨酸為脅迫/誘導(dǎo)后主要增加的蛋白質(zhì)類(lèi)產(chǎn)物,在提高EPS重金屬吸附性能中發(fā)揮關(guān)鍵作用,SMP雖熒光強(qiáng)度高,含量多[27],但是從Na2SO4-EPS和Na2S2O3-EPS的重金屬吸附能力變化上分析,SMP并不是發(fā)揮主要作用組分.
2.5 脅迫/誘導(dǎo)前后EPS傅里葉紅外光譜分析
由圖5可知,EPS在外源硫脅迫/誘導(dǎo)前后的關(guān)鍵官能團(tuán)變化明顯,脅迫后原有特征峰峰值變大,且增加了新特征峰.數(shù)學(xué)模型證明,譜峰強(qiáng)度與樣品中官能團(tuán)的濃度或數(shù)量具有正相關(guān)性[28],脅迫/誘導(dǎo)后EPS中官能團(tuán)濃度均大量增加.
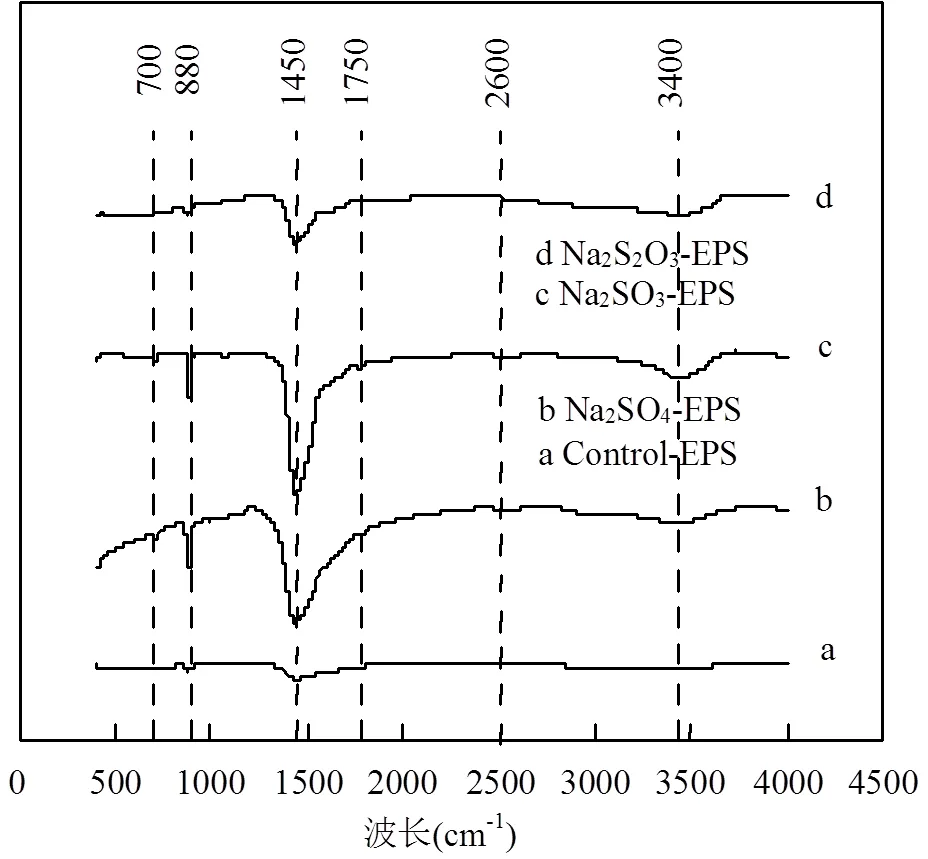
圖5 外源硫脅迫/誘導(dǎo)前后EPS傅里葉紅外光譜圖
在3400cm-1處的特征峰對(duì)應(yīng)于碳水化合物中-OH官能團(tuán)的拉伸和彎曲[29],該官能團(tuán)在Na2SO3-EPS中的相對(duì)峰值最大,Na2S2O3-EPS次之;1750cm-1處對(duì)應(yīng)于酯類(lèi)中的C=O[30],僅出現(xiàn)在Na2SO3-EPS中;1450cm-1處對(duì)應(yīng)可取代的CO32-官能團(tuán),CO32-官能團(tuán)的拉伸和彎曲可能與該官能團(tuán)在有機(jī)相中連接著-OH和-NH有關(guān)[31],該官能團(tuán)在Na2SO3-EPS中的峰值最大,Na2SO4-EPS次之; 880cm-1處為多糖中醚基(C-O-C)[32],該官能團(tuán)在Na2SO3-EPS與Na2SO4-EPS中的峰值幾乎一樣;700cm-1處為碳骨架中的-CH2[33],2600cm-1處難以確定具體的官能團(tuán)和振動(dòng)種類(lèi)[34].
上述官能團(tuán)中的-OH、C=O、C-O-C在重金屬吸附中能發(fā)揮重要作用[35],這些官能團(tuán)在Na2SO3脅迫/誘導(dǎo)后增加幅度最大,說(shuō)明Na2SO3-EPS具有最強(qiáng)的重金屬吸附能力,這與前面的試驗(yàn)和測(cè)試結(jié)果一致.
值得指出的是,2.4中類(lèi)酪氨酸的熒光強(qiáng)度變化規(guī)律與CO32-官能團(tuán)紅外光譜譜峰值變化規(guī)律一致,結(jié)合CO32-官能團(tuán)在1450cm-1的振動(dòng)規(guī)律,推斷CO32-的出現(xiàn)極有可能是其連接著類(lèi)酪氨酸中的-OH或-NH.因此,CO32-的峰值也能說(shuō)明Na2SO3- EPS含有更多的類(lèi)酪氨酸.
2.6 脅迫/誘導(dǎo)前后EPS X射線(xiàn)光電子能譜分析
2.6.1 XPS C譜分析 C是構(gòu)成EPS的主要元素之一,脅迫/誘導(dǎo)前后EPS中C元素的存在形態(tài)及相對(duì)含量如圖6和表2所示(XPS表征分析中EPS均未吸附重金屬).
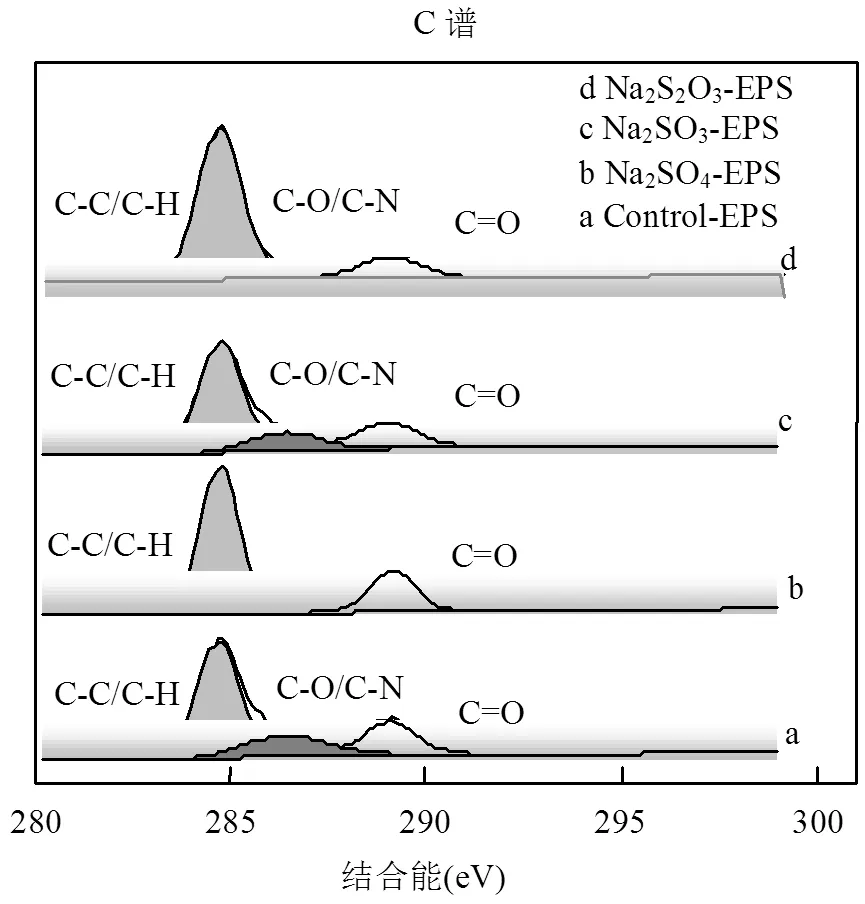
圖6 外源硫脅迫/誘導(dǎo)前后EPS的X射線(xiàn)能譜分析(C譜)
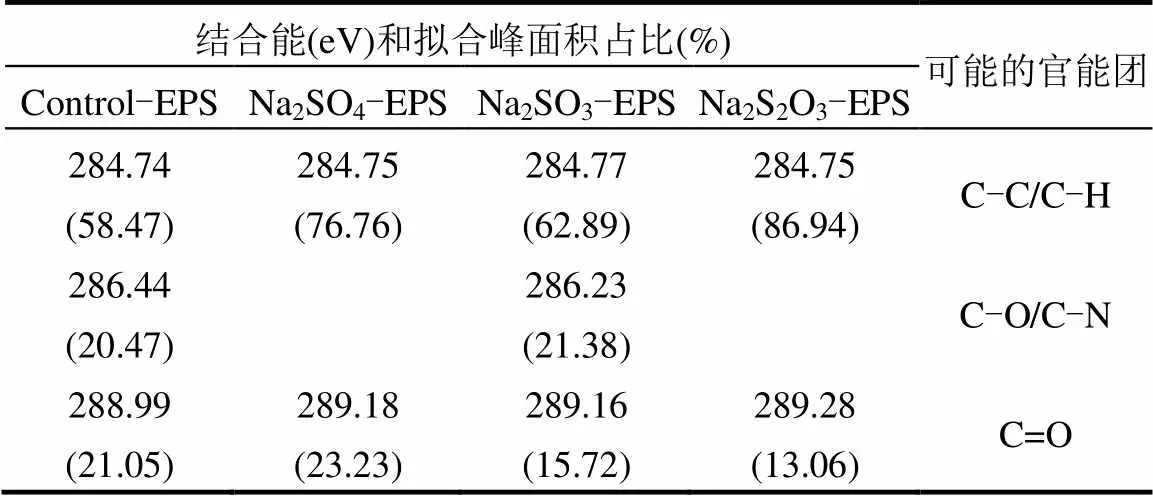
表2 脅迫/誘導(dǎo)前后EPS中C譜信息
通過(guò)圖6和表2可知,EPS中C元素的存在形態(tài)要分為3種[36]:位于284.75eV的C-C/C-H鍵,存在于脂肪族或氨基酸側(cè)鏈中[37];286.23-286.44eV處的C-O/C-N鍵;288.18-289.28處的C=O鍵.Na2SO4、Na2SO3和Na2S2O3·5H2O對(duì)C元素的脅迫/誘導(dǎo)效應(yīng)整體上一致,都合成了更多的脂肪族或氨基酸側(cè)鏈,生成更多的蛋白質(zhì).進(jìn)一步分析,脅迫后Na2SO3- EPS脂肪族或氨基酸側(cè)鏈占比最少,而C-O/C-N增加最多(其他兩種情形未測(cè)出),C=O增加不是最多的.與其吸附能力相聯(lián)系,整體上可以推斷C=O與脂肪族或氨基酸側(cè)鏈在吸附中很可能并不發(fā)揮主要作用,C-O/C-N才是吸附作用的主要官能團(tuán).官能團(tuán)與重金屬之間的作用還受到主鏈和相鄰官能團(tuán)的影響,還需要深入研究.
2.6.2 XPS O譜分析 O是構(gòu)成EPS的主要元素之一,脅迫/誘導(dǎo)前后EPS中O元素的存在形態(tài)及相對(duì)含量如圖7和表3所示.
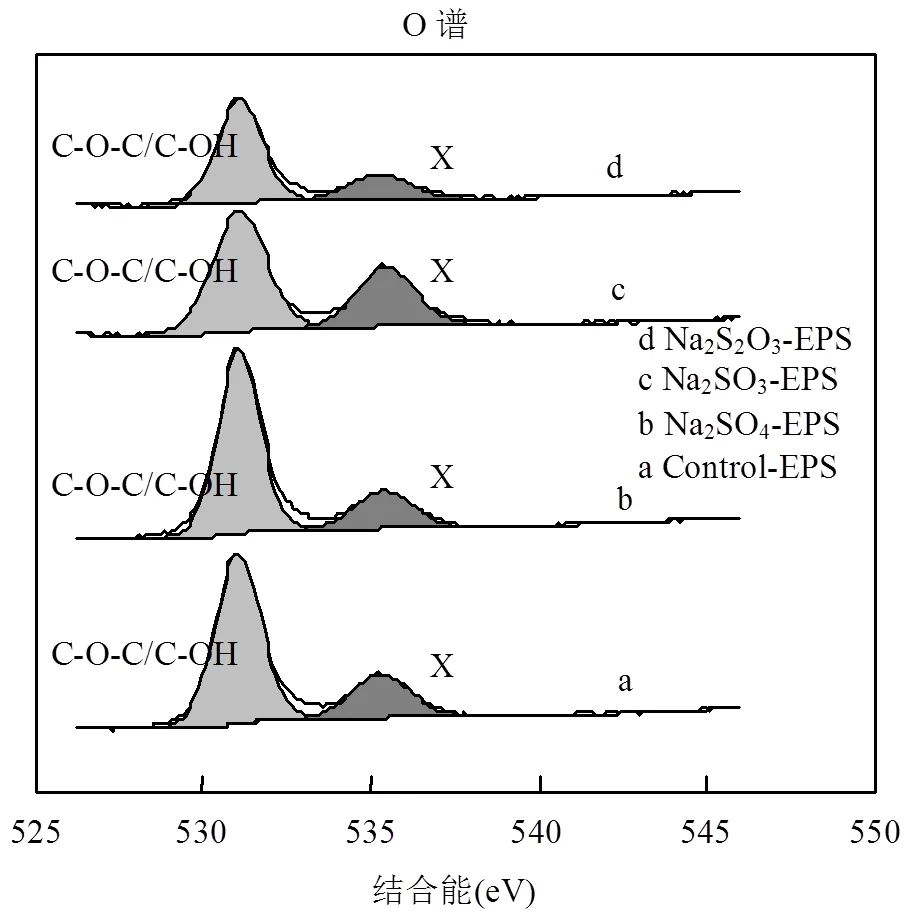
圖7 外源硫脅迫/誘導(dǎo)前后EPS的X射線(xiàn)能譜分析(O譜)
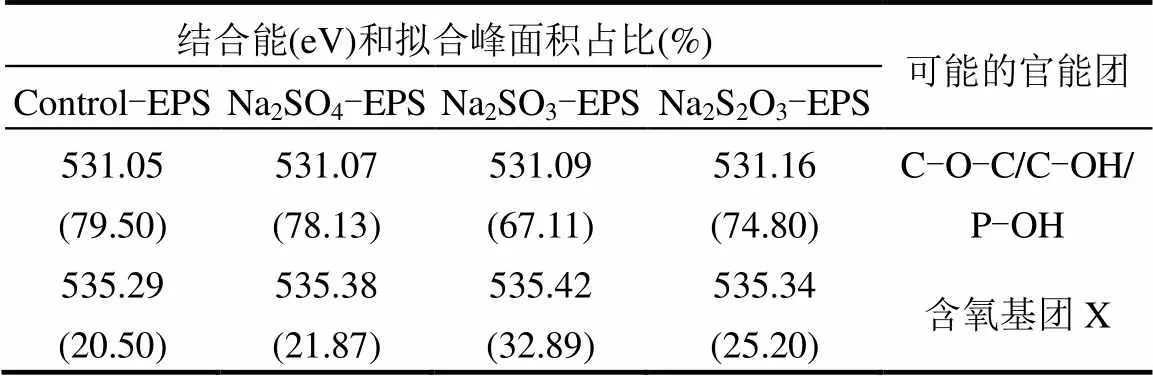
表3 脅迫/誘導(dǎo)前后EPS中O譜信息
文獻(xiàn)認(rèn)為,EPS中O的化學(xué)存在形態(tài)主要分為2種,分別對(duì)應(yīng)約531eV(C-O-C/C-OH)和約532eV (C=O)[7,38-39],但在由上述圖表中,未得到532eV處的峰,卻意外得到了535.40eV處的峰,說(shuō)明O還存在其他形態(tài),但無(wú)法通過(guò)文獻(xiàn)確定其名稱(chēng),暫記為含氧基團(tuán)X.由于其結(jié)合能較高,說(shuō)明其比較穩(wěn)定,且與重金屬有良好的結(jié)合能力,該基團(tuán)的名稱(chēng)、性質(zhì)還需要進(jìn)一步的研究.通過(guò)對(duì)比發(fā)現(xiàn),C-O-C/C-OH鍵在Na2SO3和Na2S2O3·5H2O脅迫/誘導(dǎo)后出現(xiàn)不同程度的降低,而含氧基團(tuán)X增加趨勢(shì)與Zn(Ⅱ)吸附能力變化相一致,可能在吸附中發(fā)揮主要作用.
2.6.3 XPS N譜分析 N是構(gòu)成EPS的重要元素,脅迫/誘導(dǎo)前后EPS中N元素的存在形態(tài)及相對(duì)含量如圖8和表4所示.
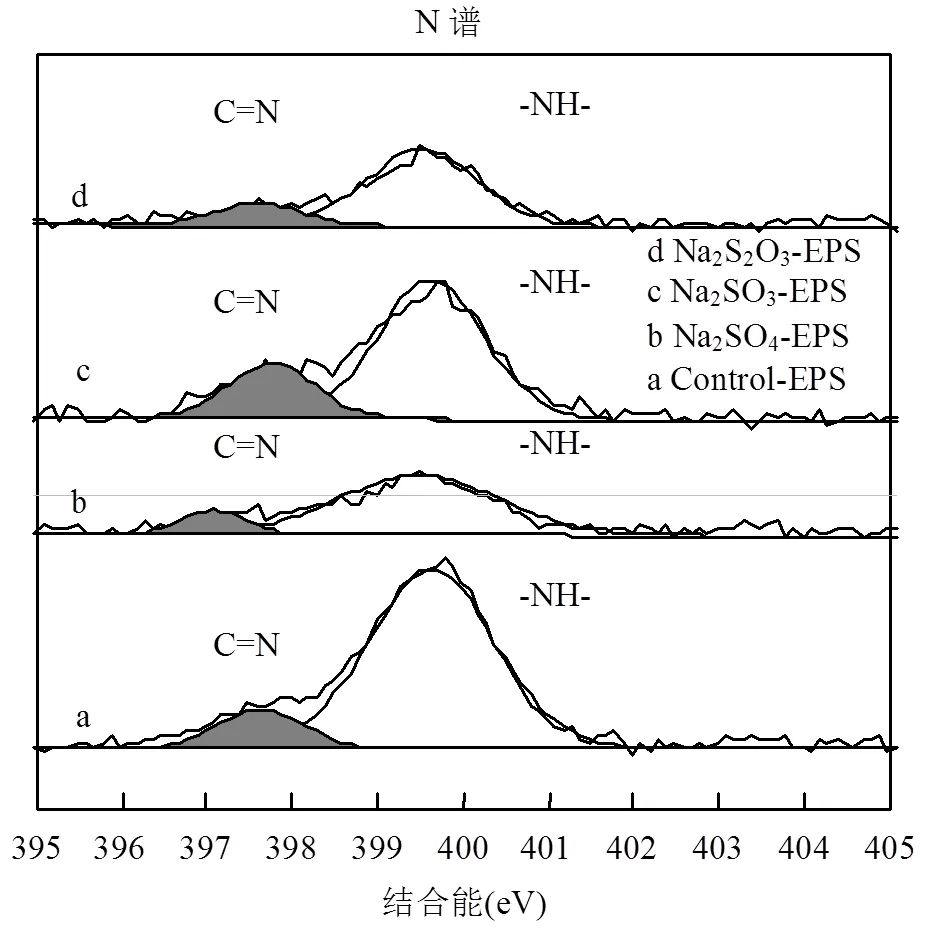
圖8 外源硫脅迫/誘導(dǎo)前后EPS的X射線(xiàn)能譜分析(N譜)
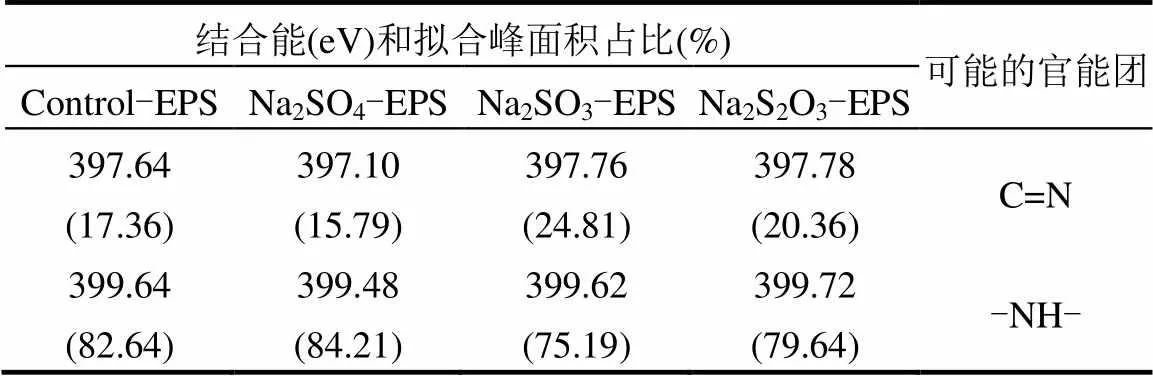
表4 脅迫/誘導(dǎo)前后EPS中N譜信息
EPS中N的化學(xué)形態(tài)主要分為2種[36]:位于397.10~397.78eV的亞胺(C=N)鍵;399.48~399.72eV處的-NH-鍵.通過(guò)對(duì)比脅迫/誘導(dǎo)前后發(fā)現(xiàn),在Na2SO3和Na2S2O3·5H2O的脅迫/誘導(dǎo)作用下C=N鍵的相對(duì)含量升高,而-NH-鍵數(shù)量降低.在Na2SO4脅迫/誘導(dǎo)作用下,兩者相對(duì)含量變化輕微.
C=N鍵的形成一般認(rèn)為是N取代了C=O鍵中的O,與重金屬螯合的能力很強(qiáng)[40-41],使得EPS的重金屬吸附能力增強(qiáng);-NH-鍵的減少說(shuō)明取代O的N可能來(lái)自-NH-鍵.Na2SO3-EPS中C=N鍵含量大幅增加很可能是重金屬吸附能力提高的又一關(guān)鍵因素.
3 結(jié)論
3.1 在一定范圍內(nèi),隨著外源硫濃度的增大,.sp.EPS產(chǎn)量呈現(xiàn)出先增加后減少的趨勢(shì).0.50g/L Na2SO3的脅迫/誘導(dǎo)效果最好,單位質(zhì)量微生物EPS和蛋白質(zhì)產(chǎn)量比脅迫/誘導(dǎo)前提高了325.66%和318.33%,達(dá)到2104.39mg/g VSS和1888.52mg/g VSS.
3.2 脅迫/誘導(dǎo)下的EPS吸附Zn(Ⅱ)的性能也發(fā)生了重大變化.EPS對(duì)Zn(Ⅱ)的吸附量在整體上均體現(xiàn)出先增加后減少的趨勢(shì).在最佳Na2SO3脅迫/誘導(dǎo)強(qiáng)度下,吸附量從脅迫/誘導(dǎo)前的481.6mg/g EPS增加到954.4mg/g EPS,提高了98.17%.
3.3 3D-EEM和FTIR分析表明,脅迫/誘導(dǎo)后EPS中可溶性微生物產(chǎn)物和類(lèi)酪氨酸的含量和與重金屬離子結(jié)合的官能團(tuán)(-OH、C=O、C-O-C等)大幅增加;XPS測(cè)試表明,Na2SO3-EPS具有豐富的C-O/ C-N、C=N和某種含氧基團(tuán),是吸附性能大幅增加的根本原因.
[1] Shi Y, Huang J, Zeng G, et al. Exploiting extracellular polymeric substances (EPS) controlling strategies for performance enhancement of biological wastewater treatments: An overview [J]. Chemosphere, 2017,180:396-411.
[2] Siddharth T, Sridhar P, Vinila V, et al. Environmental applications of microbial extracellular polymeric substance (EPS): A review [J]. Journal of Environmental Management, 2021,287:112307.
[3] Xie Q, Liu N, Lin D, et al. The complexation with proteins in extracellular polymeric substances alleviates the toxicity of Cd (II) to Chlorella vulgaris [J]. Environmental Pollution, 2020,263(Pt A):114102.
[4] Zhang P, Chen Y P, Peng M W, et al. Extracellular polymeric substances dependence of surface interactions of Bacillus subtilis with Cd(2+) and Pb(2+): An investigation combined with surface plasmon resonance and infrared spectra [J]. Colloids And Surfaces B -Biointerfaces, 2017,154:357-364.
[5] Cabral L, Giovanella P, Kerlleman A, et al. Impact of selected anions and metals on the growth and in vitro removal of methylmercury by Pseudomonas putida V1 [J]. International Biodeterioration & Biodegradation, 2014,91:29-36.
[6] 孫夢(mèng)格,宋衛(wèi)鋒,楊佐毅,等.Cd(Ⅱ)脅迫/誘導(dǎo)下銅綠假單胞菌EPS組分變化及其吸附性能 [J]. 環(huán)境科學(xué)學(xué)報(bào), 2021,41(9):3427-3436.
Sun M G, Song W F, Yang Z Y, et al.Effect of Cd(Ⅱ) stress on the variation in extracellular polymeric substances composition and adsorption performance of[J]. Journal of Environmental Science, 2021,41(9):3427-3436.
[7] Lian Z, Yang Z, Song W, et al. Effects of different exogenous cadmium compounds on the chemical composition and adsorption properties of two gram-negative bacterial EPS [J]. Science of Total Environment, 2021,806(Pt 1):150511.
[8] 曾嶠婧,周 鑫,黃 超,等.白腐菌聯(lián)合納米零價(jià)鐵強(qiáng)化去除水中Cd(Ⅱ) [J]. 中國(guó)環(huán)境科學(xué), 2022,42(7):3174-3183.
Zeng Q J, Zhou X, Huang C, et al. Enhanced removal of Cd(Ⅱ) from aqueous solution by nanoscale zero-valent iron coupled with white rot fungus [J]. China Environmental Science, 2022,42(7):3174-3183.
[9] Li Y P, You L X, Yang X J, et al. Extrapolymeric substances (EPS) in Mucilaginibacter rubeus P2displayed efficient metal(loid) bio- adsorption and production was induced by copper and zinc [J]. Chemosphere, 2022,291(Pt 1):132712.
[10] Luo X, Zhou X, Peng C, et al. Bioreduction performance of Cr(VI) by microbial extracellular polymeric substances (EPS) and the overlooked role of tryptophan [J]. Journal of Hazardous Materials, 2022,433.
[11] Yue Z B, Li Q, Li C C, et al. Component analysis and heavy metal adsorption ability of extracellular polymeric substances (EPS) from sulfate reducing bacteria [J]. Bioresource Technology, 2015,194:399- 402.
[12] 李明明.硫酸鹽還原菌胞外聚合物與金屬離子的交互作用 [D]. 合肥:合肥工業(yè)大學(xué), 2014.
Li M M. Analysis of the interaction between heavy metal and EPS isolated from sulfate reducing bacteria [D]. Hefei: Hefei University of Technology, 2014.
[13] Miqueleto A P, Dolosic C C, Pozzi E, et al. Influence of carbon sources and C/N ratio on EPS production in anaerobic sequencing batch biofilm reactors for wastewater treatment [J]. Bioresource Technology, 2010,101(4):1324-1330.
[14] Qian L, Ye X, Xiao J, et al. Nitrogen concentration acting as an environmental signal regulates cyanobacterial EPS excretion [J]. Chemosphere, 2022,291(Pt 2):132878.
[15] Li Q, Song W, Sun M, et al. Response of Bacillus vallismortis sp. EPS to exogenous sulfur stress/ induction and its adsorption performance on Cu(II) [J]. Chemosphere, 2020,251:126343.
[16] Han X, Wang Z, Chen M, et al. Acute Responses of Microorganisms from Membrane Bioreactors in the Presence of NaOCl: Protective Mechanisms of Extracellular Polymeric Substances [J]. Environmental Science & Technology, 2017,51(6):3233-3241.
[17] Dai M, Zhou G, Ng H Y, et al. Diversity evolution of functional bacteria and resistance genes (CzcA) in aerobic activated sludge under Cd(II) stress [J]. Journal of Environmental Management, 2019,250: 109519.
[18] Xu R, Fu Y, Xu Y, et al. Comparing biotransformation of extracellular polymeric substances (EPS) under aerobic and anoxic conditions: Reactivities, components, and bacterial responses [J]. Chemosphere, 2022,296:133996.
[19] Zheng Y, Bu N-S, Long X-E, et al. Sulfate reducer and sulfur oxidizer respond differentially to the invasion of Spartina alterniflora in estuarine salt marsh of China [J]. Ecological Engineering, 2017,99: 182-190.
[20] 王 原.珠江沉積物中SRB的群落結(jié)構(gòu)、分離篩選和生理生化特性鑒定 [D]. 華南理工大學(xué), 2013.
Wang Y. Community Structure, Isolation, Biochemical and Physiological Identification of Sulfate-Reducing Bacteria from Pearl River Sediments [D]. Guangzhou: South China University of Technology, 2013.
[21] Su Z, Li X, Xi Y, et al. Microbe-mediated transformation of metal sulfides: Mechanisms and environmental significance [J]. Science of Total Environment, 2022,825:153767.
[22] Wang J, Li Q, Li M M, et al. Competitive adsorption of heavy metal by extracellular polymeric substances (EPS) extracted from sulfate reducing bacteria [J]. Bioresource Technology, 2014,163:374-6.
[23] Qi S, Zhang M, Guo X, et al. Controlled extracellular biosynthesis of ZnS quantum dots by sulphate reduction bacteria in the presence of hydroxypropyl starch as a mediator [J]. Journal of Nanoparticle Research, 2017,19(6).
[24] 萬(wàn)正強(qiáng).EPS在硫酸鹽脫硫弧菌(Desulfovibrio desulfuricans)去除重金屬Cd2+過(guò)程作用研究 [D]. 合肥:合肥工業(yè)大學(xué), 2013.
Wan Z Q. Study on the role of EPS in the removal of heavy metal Cd2+by Desulfovibrio desulfuricans [D]. Hefei: Hefei University of Technology, 2013.
[25] Zan F, Huang H, Guo G, et al. Sulfite pretreatment enhances the biodegradability of primary sludge and waste activated sludge towards cost-effective and carbon-neutral sludge treatment [J]. Science of Total Environment, 2021,780:146634.
[26] Yu B, Lou Z, Zhang D, et al. Variations of organic matters and microbial community in thermophilic anaerobic digestion of waste activated sludge with the addition of ferric salts [J]. Bioresource Technology, 2015,179:291-298.
[27] Ma B, Li S, Wang S, et al. Effect of Fe3O4nanoparticles on composition and spectroscopic characteristics of extracellular polymeric substances from activated sludge [J]. Process Biochemistry, 2018,75:212-220.
[28] Palencia M. Functional transformation of Fourier-transform mid-infrared spectrum for improving spectral specificity by simple algorithm based on wavelet-like functions [J]. Journal of Advanced Research, 2018,14: 53-62.
[29] Xu Y, Liu P, Zhang Y. Mid-infrared spectroscopy of hemispherical water droplets [J]. Spectrochimica Acta Parta A-Molecular And Biomolecular Spectroscopy, 2022,264:120256.
[30] Kamnev A A, Tugarova A V, Dyatlova Y A, et al. Methodological effects in Fourier transform infrared (FTIR) spectroscopy: Implications for structural analyses of biomacromolecular samples [J]. Spectrochimica Acta Parta A-Molecular And Biomolecular Spectroscopy, 2018,193:558-564.
[31] Waheed S, Sultan M, Jamil T, et al. Comparative Analysis of Hydroxyapatite Synthesized by Sol-gel, Ultrasonication and Microwave Assisted Technique [J]. Materials Today: Proceedings, 2015,2(10):5477-5484.
[32] Ali H U, Iqbal D N, Iqbal M, et al. HPMC crosslinked chitosan/ hydroxyapatite scaffolds containing Lemongrass oil for potential bone tissue engineering applications [J]. Arabian Journal of Chemistry, 2022,103850.
[33] Khare N, Bajpai J, Bajpai A K. Graphene coated iron oxide (GCIO) nanoparticles as efficient adsorbent for removal of chromium ions: Preparation, characterization and batch adsorption studies [J]. Environmental Nanotechnology, Monitoring & Management, 2018, 10:148-162.
[34] Bao F, Zong L, Li N, et al. Synthesis of novel poly(phthalazinone fluorenyl ether ketone ketone)s with improved thermal stability and processability [J]. Thermochimica Acta, 2020,683.
[35] Zhang M, Hou S, Li Y, et al. Single evaluation and selection of functional groups containing N or O atoms to heavy metal adsorption: Law of electric neutrality [J]. Chemosphere, 2022,287(Pt 2):132207.
[36] Maaza L, Djafri F, Belmokhtar A, et al. Evaluation of the influence of Al2O3nanoparticles on the thermal stability and optical and electrochemical properties of PANI-derived matrix reinforced conducting polymer composites [J]. Journal of Physics and Chemistry of Solids, 2021,152.
[37] 連澤陽(yáng),楊佐毅,宋衛(wèi)鋒,等.外源Cd(Ⅱ)脅迫Alcaligenes faecalis過(guò)程中陰離子對(duì)EPS產(chǎn)量及其特性的影響 [J]. 環(huán)境科學(xué)學(xué)報(bào), 2022,42(4):81-90.
Lian Z Y, Yang Z Y, Song W F, et al. The effect of anions on the yield and characteristics of EPS during the process of exogenous Cd(Ⅱ) stress/induction of Alcaligenes faecalis [J]. Journal of Environmental Science: 2022,42(4):81-90.
[38] Szcze? A, Czemierska M, Jarosz-Wilko?azka A. Calcium carbonate formation on mica supported extracellular polymeric substance produced by Rhodococcus opacus [J]. Journal of Solid State Chemistry, 2016,242:212-221.
[39] Lin D, Ma W, Jin Z, et al. Interactions of EPS with soil minerals: A combination study by ITC and CLSM [J]. Colloids And Surfaces B -Biointerfaces, 2016,138:10-6.
[40] Han J, Pei L, Du Y, et al. Tripolycyanamide-2,4,6-triformyl pyrogallol covalent organic frameworks with many coordination sites for detection and removal of heavy metal ions [J]. Journal of Industrial and Engineering Chemistry, 2022,107:53-60.
[41] Qin L, Ge Y, Deng B, et al. Poly (ethylene imine) anchored lignin composite for heavy metals capturing in water [J]. Journal of the Taiwan Institute of Chemical Engineers, 2017,71:84-90.
The EPS characteristics ofsp. and its adsorption performance for Zn(Ⅱ) under exogenous sulfur induction.
GAN Yu, SONG Wei-feng*, YANG Zuo-yi, LIAN Ze-yang, MA Shuang-nian, HUANG Xiang-wu, YANG Ren-gao, WEN Yan-biao
(School of Environmental Science and Engineering, Guangdong University of Technology, Guangzhou 510006, China)., 2022,42(11):5144~5152
In this paper, the effect of exogenous sulfur (Na2SO4, Na2SO3, Na2S2O3·5H2O) stress/induction on extracellular polymeric substances(EPS) ofsp. (.sp.)was studied. The results showed that addition of 0.50g/L Na2SO3led to the highest EPS yield of 2104.39mg/g VSS (1888.52mg/g VSS of protein content), which was 300% higher than that without Na2SO3addition, and as a result, 98.17% increase in Zn(II) adsorption capacity (954.4mg/g EPS) achieved. Three-dimensional fluorescence (3D-EEM) spectral results showed that tyrosine-like substances in EPS were greatly increased aftersulfurstress/induction; Fourier transform infrared spectroscopy (FTIR) results showed that significant increases in functional groups such as —OH, C=O, C—O—C EPS were mainly responsible for the enhance adsorption of Zn(II); The results of X-ray photoelectron spectroscopy (XPS) results showed that the content of C—O/C—N, C=N and oxygen-containing functional group (X) in EPS increased after sulfur stress/induction (Na2SO3and Na2S2O3·5H2O).
exogenous sulfur;stress/induction;EPS;Zn(Ⅱ)
X172
A
1000-6923(2022)11-5144-09
甘 雨(1997-),男,廣東惠州人,廣東工業(yè)大學(xué)碩士研究生,主要從事EPS吸附重金屬及介導(dǎo)合成金屬硫化物.發(fā)表論文2篇.
2022-04-18
廣東省自然科學(xué)基金資助項(xiàng)目(2021A1515010558)
* 責(zé)任作者, 教授, weifengsong@gdut.edu.cn

