離子對化合物[(IBz)2Im](TCNQ)的合成與結構表征
陳友存 王芳芳 王 彥 汪快兵
(安慶師范學院安徽省功能配合物重點實驗室,安慶 246011)
研究簡報
離子對化合物[(IBz)2Im](TCNQ)的合成與結構表征
陳友存*王芳芳 王 彥 汪快兵
(安慶師范學院安徽省功能配合物重點實驗室,安慶 246011)
TCNQ;晶體結構;離子對化合物
The exploration for new compounds with enchanting electronic,magnetic and photomagnetic properties has prompted chemists to combine different spin carriers within the same molecular or supramolecular entity[1-3].One of the most extensively used radicals in these studies is the planar organic molecule TCNQ.The planar organic molecule TCNQ(7,7,8,8-tetracyanoquinodimethanide,[C8H4(CN)4])is a suitable acceptor in charge-transferprocesses,which shows extended electrical structures that are similar to[M(mnt)2]-(M=Ni,Pd and Pt,mnt=Maleonitriledithiolato)monoanion.The TCNQ-has been extendedly employed to build molecular solids with low-dimensional conducting and magnetic feature,in which the electronic transport and magnetic coupled interactions can be achieved through π-π interactions between the radical along the stacking direction[4-7].In our previous study,we employed benzylpyridinium derivatives([RBzPy]+,where R represents a substituent group)with flexible molecular configurations as counter-cations to control the arrangement of[M(mnt)2]-monoanions(M=Ni,Pd and Pt)to achieve a series of molecular solids with segregated stacks of cations and anions[8-10].
As a result,the size and shape of counter-cationsplay an important role in determining the ground-state properties of the resulting materials.Thus,these characteristics of TCNQ-radical ion have encouraged us to expand our research,studying the series of molecular solids in order to gain more insight into the relationship between the intermolecular cooperation interactions and the magnetic properties with lowdimensional structural features.Up to now,we have focused our attention on the construction of novel ionpair compounds by changing the proportion of cation and anion,and the results showed that the change of molar ratio leads anion to exhibit different packing structures.In this paper,we report the synthesis and crystal structure of a novel ion-pair complex,namely,[(IBz)2Im](TCNQ),and its IR spectra are also discussed.
1 Experimental
1.1 Physical measurements
Imidazole and iodobenzyl bromide were purchased from Acros and used without further purification,and all of the other reagents were commercially available and used as received without purification.The FTIR spectrum was recorded on an Avatar 360 FTIR spectrometer by using a KBr disk.Elemental analyses of C,H and N were carried out on a Vario ELⅢElemental Analyzer.
1.2 Synthesis of[(IBz)2Im]Br
1∶2 Molar equivalent of imidazole and iodobenzyl bromide were mixed directly in acetone solution,and the mixture was refluxed for 3 h.The white microcrystalline products formed were filtered off,washed with acetone and dried in vacuum[11].Yield:62.8%.
1.3 Synthesis of[(IBz)2Im][TCNQ]
The[(IBz)2Im]Br and LiTCNQ in 1∶1 mol ratio were directly mixed in a solution of CH3OH,and then the mixture was refluxed for 5 h.The purple-black microcrystalline products formed were filtered off,then washed with CH3OH and dried in vacuum.Yield:59.6%.The purple-black products were dissolved in acetonitrile and single crystals suitable for X-ray analysis were obtained by dispersing Et2O into the CH3CN solution for 2 weeks.Anal.Calcd.for C29H19I2N6(%):C,49.39;N,11.92;H,2.72.Found(%):C,49.65;N,11.56;H,3.01.IR(cm-1):3 446(m),2185(w),1 585(m),1 505(s),1 350(m),1178(s),822(s),721(s).
1.4 Crystal structure determination
A crystal with dimensions of 0.10 mm×0.15 mm×0.15 mm was selected for data collection at 293(2)K by using a Bruker Smart ApexⅡCCD equipped with a Mo Kα radiation (λ=0.071 073 nm).A total of 6 052 reflections were collected in the range of 2.20°<θ<25.26°with 4 025 independent ones(Rint=0.082 0),of which 4025 were observed with I>2σ(I)and used in the structure solution and refinement.The final R=0.053 2,wR=0.1112(w=1/[σ2(Fo2)+(0.0374P)2+1.3395P],where P=(Fo2+2Fc2)/3,S=1.022,(Δ/σ)max=0.001,(Δρ)max=918 e·nm-3and (Δρ)min=-961 e·nm-3).The structure was solved by direct methods and refined by full-matrix least-squares techniques (SHELXTL-97)[12].The experimental X-ray data for the title compound and the selected bond lengths are listed in Tables 1 and 2,respectively.The hydrogen bond lengths and bond angles are listed in Table 3.
CCDC:759523.

Table 1 Crystallographic data for the complex
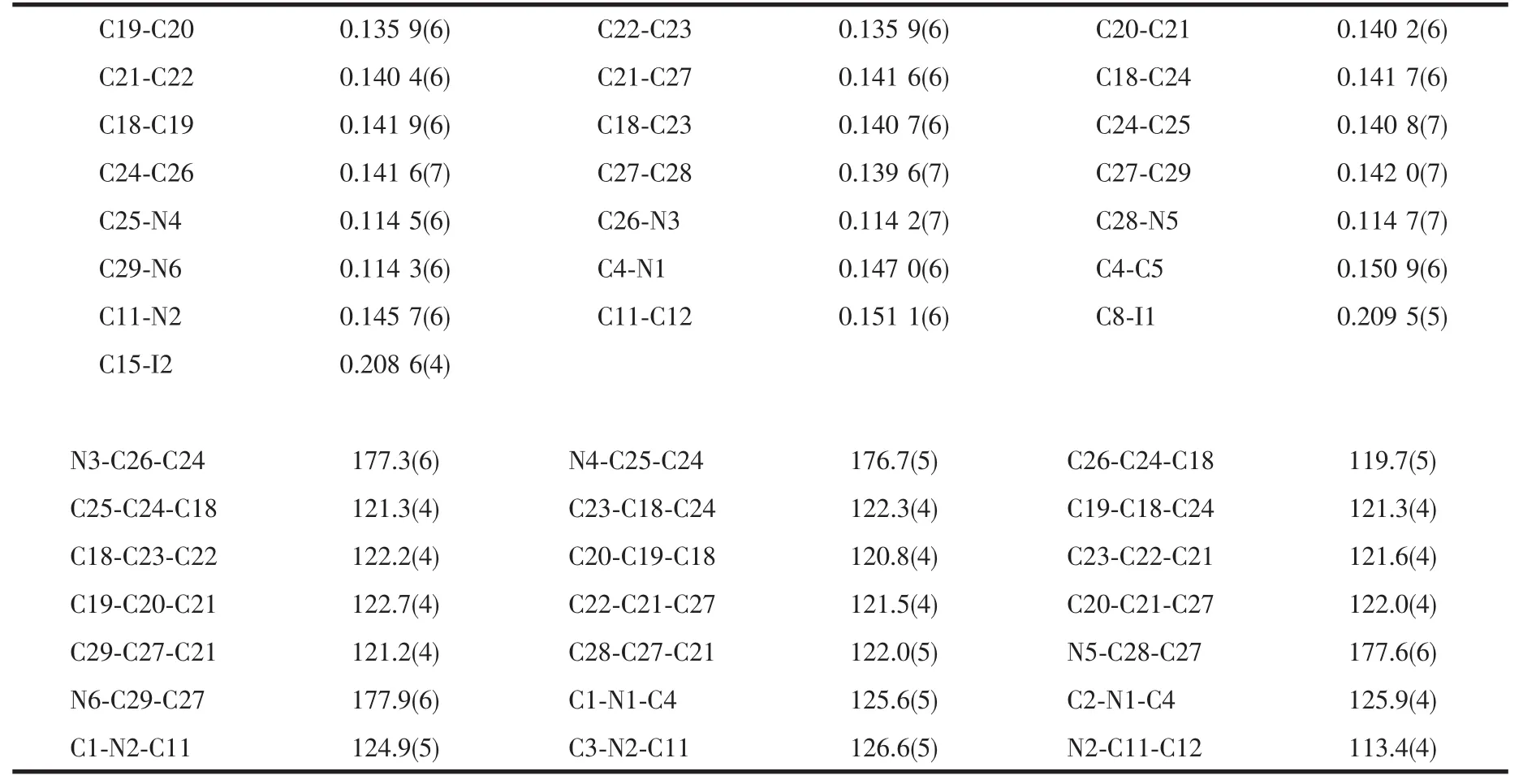
Table 2 Selected bond lengths(nm)and bond angles(°)

Table 3 Hydrogen bond lengths and bond angles
2 Results and discussion
2.1 Crystal structure of the complex
The title compound crystallizes in a monoclinic system with space group P21/c.As shown in Fig.1,the compound consists of one [(IBz)2Im]+cation and one independent TCNQ anions in an asymmetric unit.The averaged lengths of chemically similar bonds in the TCNQ molecule were listed in Table 2,which are very close to those in other reported compounds containing TCNQ-[13].The centroid-to-centroid distances of the neighboring TCNQs are 0.327 4(3)nm(Fig.2),indicating the strong π-π interactions between the TCNQ-.The TCNQ-anions are stacked into column with isolated πdimers.
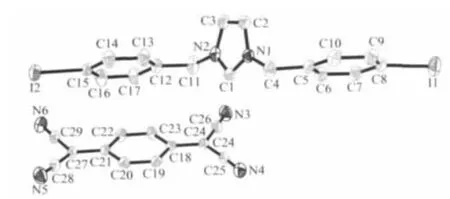
Fig.1 A fragment of the title compound with 30%thermal ellipsoids
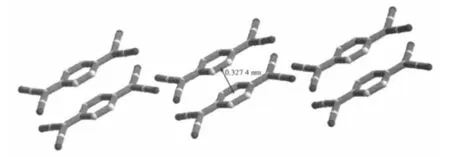
Fig.2 A side-view of the one-dimensional anionic chain of TCNQ-,hydrogen atoms were omitted for clarity
The[(IBz)2Im]+cation has a Z-shaped conformation,the dihedral angles between the rings and the reference plane(C5/C4/N1)are 140.0°for benzene ring(C5/C6/C7/C8/C9/C10)and 72.8°for imidazole ring,while the dihedral angles between the rings and the other reference plane(C12/C11/N2)are 1.1°for benzene ring(C12/C13/C14/C15/C16/C17)and 88.1°for imidazole ring.It is noteworthy that the two neighboring[(IBz)2Im]+cations are dimerized in chair-like conformation and stabilized by C-H…I hydrogen bonds between the adjacent cations(Fig.3).TCNQ-columns and[(IBz)2Im]+columns are held together via C-H…N hydrogen bonding interactions to stabilize the whole structure(Fig.4).The most prominent structural features of the title compound are the completely segregated stacking columns of the TCNQ-anions and [(IBz)2Im]+cations.The result shows that the noncovalent interactions play an important role in stabilizing the architectures in selfassembly process.
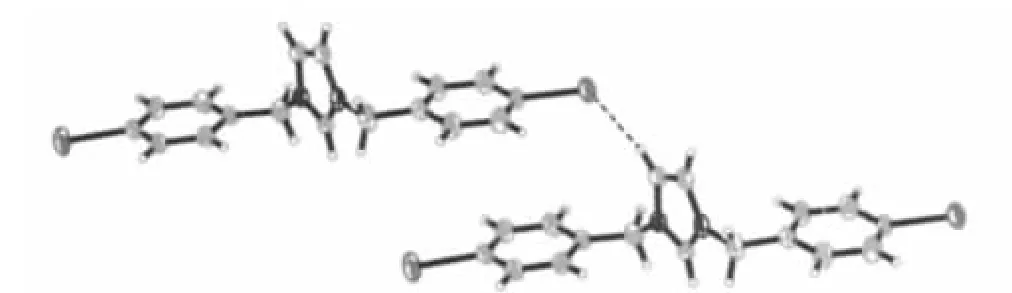
Fig.3 A side-view of two adjacent cations,exhibit hydrogen bonding with broken line
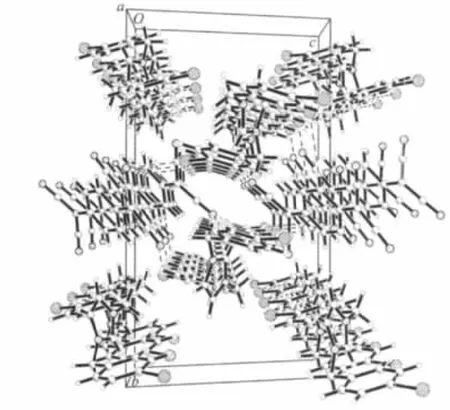
Fig.4 Packing diagram for the compound along the a axis
2.2 IR spectrum of the title complex
In order to investigate and prove the valence of TCNQ species in the title complex,the IR spectrum complex has been investigated. Since the ν(C≡N)band energies roughly provide general information on the extent of reduction of TCNQ0(>2 220 cm-1),TCNQ-(2 100 ~2 220 cm-1)or TCNQ2-(<2 100 cm-1),the vibration of ν(C≡N)often used to identify the form of TCNQ[14].The most significative vibration frequencies of the TCNQ0and TCNQ-are listed in Table 4[15].Comparison with the data of neutral TCNQ and TCNQ-indicates that TCNQ-is presented in the compound,as noted by the shifts of the nitrile frequencies to lower energies(from 2 228 to 2 185 cm-1for compound)[16-17].As mentioned above,the analysis of IR spectrum is consistent with the results of crystal structure analysis.

Table 4 Most significative vibration frequencies(cm-1)of the TCNQ units
[1]Madalan A M,Roesky H W,Andruh M,et al.Chem.Commun.,2002:1638-1639
[2]Jiang K,Zhao B L,Wang L Y,et al.Chinese J.Struct.Chem.,2007,26:860-866
[3]Wang L Y,Wang Y F,Ma L F,et al.Chinese J.Struct.Chem.,2005,24:283-286
[4]Wang Y F,Wang L Y.Chinese J.Struct.Chem.,2007,26:1461-1465
[5]Radvakova A,Kajnakova M,Feher A,et al.Acta Phys.Polonica A,2008,113:499-502
[6]Schelter E J,Morris D E,Scott B L,et al.Inorg.Chem.,2007,46:5528-5536
[7]Mukai K,Jinno S,Shimobe Y,et al.Mater.Chem.,2003,13:1614-1621
[8]Ren X M,Chen Y C,He C,et al.J.Chem.Soc.,Dalton Trans.,2002:3915-3918
[9]Ren X M,Ma J,Lu C S,et al.J.Chem.Soc.,Dalton Trans.,2003:1345-1351
[10]Ren X M,Meng Q J,Song Y,et al.Inorg.Chem.,2002,41:5686-5692
[11]Bulgarevich S B,Bren D V,Movshovic D Y.J.Mol.Struct.,1994,317:147-159
[12]Sheldrick G M.SHELXS97 and SHELXL97,University of G?ttingen,Germany,1997.
[13]Liu G X,Ren X M,Kremer P K,et al.J.Mol.Struct.,2005,743:125-133
[14]CHEN You-Cun(陳友存),LIU Guang-Xiang(劉光祥),WANG Ming(汪 銘),et al.Spectroscopy and Spectral Analysis(GuanpuxueYuGuangpuFenxi),2005,25(3):395-397
[15]Ballester L,Gil A M,Gutierrez A,et al.Inorg.Chem.,2000,39:2837-2842
[16]Cornlissen P J,Van Dienmen J H,Groeneveld L R,et al.Inorg.Chem.,1992,31:198-202
[17]Pawlak W M,Graja A,Lequan M,et al.Inorg.Chem.,1992,26:1328-1331
Synthesis and Structure Characterization of Ion-Pair Compound[(IBz)2Im](TCNQ)
CHEN You-Cun*WANG Fang-FangWANG Yan WANG Kuai-Bing
(Anhui Key Laboratory of Functional Coordination Compounds,Anqing Normal University,Anqing,Anhui 246011)
A novel compound[(IBz)2Im](TCNQ)[(IBz)2Im=1,3-bis(4-iodobenzyl)imidazole cation,TCNQ-=7,7,8,8-tetracyanoquinodimethanide anion]was synthesized by the reaction of[(IBz)2Im]Br and LiTCNQ in CH3OH and its structure was determined by single-crystal X-ray diffraction.The crystal belongs to monoclinic,space group P21/c with a=1.14735(18)nm,b=2.0281(3)nm,c=1.2641(2)nm,β=104.73(0)°,V=2.666(65)nm3,Z=4,C29H19I2N6,Mr=705.30,Dc=1.757 g·cm-3,R1=0.0532 and wR2=0.1112.The structure analysis shows that the anions are stacked into column with isolated π-dimers,and there is one type of TCNQ entries (TCNQ-),in agreement with the IR spectra analysis of the compound.The most prominent structural features are the completely segregated stacking columns of the TCNQ-anions and[(IBz)2Im]+cations.CCDC:759523.
TCNQ;crystal structure;ion-pair compound
O613.44;O621.3
A
1001-4861(2010)06-1113-04
2010-01-04。收修改稿日期:2010-03-30。
國家自然科學基金項目(No.20771006,20901004)和安徽省教育廳重點基金項目(No.2006kj042A)資助。*
。 E-mail:chenyc@aqtc.edu.cn
陳友存,男,62歲,教授;研究方向:功能配合物。

