胰腺神經內分泌腫瘤預后模型的建立與驗證
王玉,陳慶法,劉佳,陳興田
胰腺神經內分泌腫瘤預后模型的建立與驗證
王玉,陳慶法,劉佳,陳興田
臨沂市中心醫院消化內科,山東臨沂 276400
探討影響胰腺神經內分泌腫瘤預后的危險因素,構建胰腺神經內分泌腫瘤預后預測模型。選取SEER數據庫中2004年1月至2015年12月經病理明確診斷為胰腺神經內分泌腫瘤的患者3606例,按照3∶1分割為訓練集(=2704)和驗證集(=902),在訓練集中通過Cox比例風險模型篩選影響胰腺神經內分泌腫瘤預后的危險因素,進一步構建其預后模型并繪制列線圖。分別在訓練集和驗證集中對模型的預測效能進行內部及外部驗證。單因素Cox回歸分析顯示,性別、年齡、婚姻狀態、腫瘤部位、分化程度、TNM分期、美國癌癥聯合委員會(American Joint Committee on Cancer,AJCC)分期、手術均是影響胰腺神經內分泌腫瘤預后的危險因素(<0.05);多因素Cox回歸分析顯示,年齡、性別、婚姻狀態、分化程度、TNM分期、手術均是影響胰腺神經內分泌腫瘤預后的危險因素(<0.05)。最終將年齡、性別、分化程度、腫瘤部位、TNM分期、手術、婚姻狀態等變量納入預測模型并繪制列線圖。在訓練集和驗證集中,預測模型的C指數分別為0.8579和0.8572。訓練集和驗證集中3年、5年生存率的校準曲線顯示,預測生存率與實際生存率存在較好的一致性。構建的胰腺神經內分泌腫瘤預測模型具有良好的預測價值。
胰腺神經內分泌腫瘤;預測模型;SEER數據庫
胰腺神經內分泌腫瘤起源于胰腺的神經內分泌細胞,是僅次于胰腺導管細胞癌的胰腺腫瘤,具有相對惰性的生物學行為[1],占所有胰腺腫瘤的1%~2%,占所有神經內分泌腫瘤的7.0%[2],免疫組織化學可見突觸素和(或)嗜鉻粒蛋白的陽性表達[3]。根據是否存在臨床癥狀,胰腺神經內分泌腫瘤分為功能性和無功能性,具體取決于它們是否釋放產生癥狀的激素,其中60%~90%的胰腺神經內分泌腫瘤是無功能的[4]。隨著診斷意識提高、影像學及分子病理學的進展,其發病率也在逐年提高。研究數據表明,在過去的40年里,胰腺神經內分泌腫瘤發病率約6.98/10萬,較前增長6.4倍[5]。與胰腺導管腺癌相比,胰腺神經內分泌腫瘤的特征是發病率更低,年齡更小,預后更好[6],5年生存率80%~90%[7];但胰腺神經內分泌腫瘤的臨床特征和預后高度異質性,其中一小部分患者表現出惡性特征,給臨床實踐帶來挑戰[8]。由于其異質性較強,目前關于胰腺神經內分泌腫瘤的生存預后與風險分層缺乏公認的精準分期系統[9]。本研究基于SEER數據庫提取相關發病人群的大樣本數據,分析胰腺神經內分泌腫瘤的臨床病理特征,并進一步構建和驗證其預后模型,為個體化臨床方案的制定提供參考。
1 資料與方法
1.1 資料獲取
研究數據來自SEER數據庫(https://seer.cancer. gov/),通過SEER*Stat 8.4軟件收集數據庫中2004年1月至2015年12月經病理明確診斷為胰腺神經內分泌腫瘤的患者,其中組織學類型根據《國際疾病分類腫瘤學專輯》第3版(ICD-O-3),選擇神經內分泌腫瘤,代碼為8150,8151,8152,8153,8155,8156,8240,8241,8242,8243,8246,8249。納入標準:①年齡>18歲;②經病理組織學確診為神經內分泌腫瘤/癌(代碼同上);③原發部位為胰腺。排除標準:①30d內死亡;②相關臨床信息缺失,如淋巴結轉移、遠處轉移、治療方式、隨訪時間、生存狀態等。根據上述納入、排除標準,本研究共納入3606例患者。采用隨機樣本分割(分割比3∶1)分為訓練集(=2704)與驗證集(=902)。
1.2 統計學方法
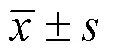
2 結果
2.1 胰腺神經內分泌腫瘤的臨床病理特征
3606例胰腺神經內分泌腫瘤患者的臨床病理特征見表1。訓練集和驗證集的年齡、性別、部位、分化程度、美國癌癥聯合委員會(American Joint Committee on Cancer,AJCC)分期、TNM分期、手術、種族、婚姻狀態及生存時間等比較差異均無統計學意義(>0.05),證明兩組數據集間具有可比性。
2.2 胰腺神經內分泌腫瘤特異性死亡的單因素及多因素Cox回歸分析
基于訓練集數據,以胰腺神經內分泌腫瘤特異性死亡作為因變量(存活定義為0,死亡定義為1),其臨床病理特征作為自變量進行單因素及多因素Cox回歸分析。單因素Cox分析結果顯示,性別、年齡、婚姻狀態、腫瘤部位、分化程度、TNM分期、AJCC分期、手術均是影響胰腺神經內分泌腫瘤預后的危險因素(<0.05)。鑒于胰腺神經內分泌腫瘤較強的異質性,T分期、N分期、M分期可能提供更多的臨床信息,AJCC分期與上述變量可能存在共線性問題,多因素Cox回歸分析未將AJCC分期納入,結果顯示,年齡、性別、婚姻狀態、分化程度、TNM分期、手術均是影響胰腺神經內分泌腫瘤預后的獨立危險因素(<0.05)。
2.3 訓練集中列線圖的構建
在訓練集中,將多因素篩選的年齡、性別、婚姻狀態、分化程度、TNM分期、手術等變量納入模型。胰腺腫瘤的發病部位是影響預后的關鍵因素,因此將腫瘤部位納入模型中,以此構建胰腺神經內分泌腫瘤3年、5年生存率預測的列線圖,見圖1。列線圖是回歸模型的可視化圖表,根據每個自變量對應的分值相加,可計算出個體的生存概率。
2.4 模型預測效能的評價
在訓練集和驗證集中,預測模型的C指數分別為0.8579和0.8572。利用列線圖在訓練集和驗證集中預測胰腺神經內分泌腫瘤3年、5年生存率的ROC曲線見圖2,訓練集中曲線下面積(area under the curve,AUC)分別為0.882、0.873,驗證集中AUC分別為0.878、0.877。訓練集和驗證集中3年、5年生存率的校準曲線顯示,預測生存率與實際生存率存在較好的一致性,見圖3。

表1 3606例胰腺神經內分泌腫瘤患者的臨床病理特征
注:*包含未婚、離婚、寡居等婚姻狀態

表2 胰腺神經內分泌腫瘤特異性死亡的單因素及多因素Cox回歸分析

續表2
注:*包含未婚、離婚、寡居等婚姻狀態

圖1 胰腺神經內分泌腫瘤3年、5年生存率的列線圖

圖2 訓練集和驗證集列線圖預測3年、5年生存率的ROC曲線
A.訓練集3年生存率;B.訓練集5年生存率;C.驗證集3年生存率;D.驗證集5年生存率

圖3 訓練集和驗證集列線圖預測3年、5年生存率的校準曲線
A.訓練集3年生存率;B.訓練集5年生存率;C.驗證集3年生存率;D.驗證集5年生存率
3 討論
與其他神經內分泌腫瘤相比,胰腺神經內分泌腫瘤異質性更強,具有獨特的病理生理學特征,給該種疾病的管理帶來一定的挑戰[9]。由于缺乏特定的生物標志物和疾病相關癥狀,大多數患者在疾病后期才被診斷出來。大多數胰腺神經內分泌腫瘤是無功能的,且診斷時腫瘤已進展至晚期不可切除或已轉移[10]。隨著影像學及各種檢測技術的發展,胰腺神經內分泌腫瘤檢出率越來越高,然而,仍然沒有一套標準可確定患者的預后[11]。鑒于上述因素,目前需要一種更精確的預后工具估計這些患者的生存率,以協助臨床決策和治療策略的優化[12]。由于具有強異質性和復雜的生物學行為,僅使用傳統的分期系統評估其預后是不精確的,必須建立一個有效的預后系統[13]。研究表明,在多種惡性腫瘤中,列線圖比經典的AJCC分期具有更好的預測能力[14]。除了臨床分期外,其他人口統計學和臨床特征,如診斷時的年齡、性別、腫瘤大小、腫瘤位置和微環境及腫瘤炎癥特征也可影響胰腺神經內分泌腫瘤患者的預后[15]。本研究基于SEER數據庫,從人口學及臨床病理特征等基本信息入手,構建胰腺神經內分泌腫瘤預后模型,同時進行外部驗證,為胰腺神經內分泌腫瘤預后評價提供臨床證據。
目前大多數情況下,手術仍是胰腺神經內分泌腫瘤的主要治療手段。所有>2cm的胰腺神經內分泌腫瘤,無論大小及功能如何,均應手術切除[16]。<2cm的胰腺神經內分泌腫瘤通常表現出良性行為,是否進行手術仍存在爭議[17]。一項單中心研究表明,對<2cm的胰腺神經內分泌腫瘤進行非手術治療是安全可行的,平均隨訪3~4年,隨訪期間未發生轉移及特異性病死率增加[18];薈萃分析顯示,即使對<2cm的胰腺神經內分泌腫瘤,手術也具有顯著的總體生存獲益[19]。本研究多因素分析顯示,手術治療可使胰腺神經內分泌腫瘤死亡風險下降73%,這與既往研究結果一致。既往研究表明,相較于胰腺體尾部,胰腺頭部腫瘤直徑更大,淋巴結轉移更常見,更容易發生局部侵襲和遠處轉移,因此胰頭部胰腺神經內分泌腫瘤預后更差[20]。本研究結果顯示,相較于其他部位,胰頭部神經內分泌腫瘤預后更差。TNM分期是常用的惡性腫瘤預后評估工具,隨著分期增加,預后越差。此外,本研究發現,男性、年齡≥60歲、婚姻狀態(非已婚)均是胰腺神經內分泌腫瘤預后不良的危險因素,與既往研究結果一致[21-22]。
本研究仍有一定局限性。首先,本研究為基于SEER數據庫的回顧性研究,建立的預測模型仍需在人群中進一步行前瞻性驗證;其次,SEER數據庫中并沒有關于胰腺神經內分泌腫瘤的分子病理信息,且缺乏手術及術后并發癥、放化療等具體信息,因此納入變量有進一步調整的可能。未來仍需要大樣本、前瞻性、更加詳細的臨床病理特征構建理想的預測模型,以更好地指導胰腺神經內分泌腫瘤的臨床診治。
[1] HALFDANARSON T R, RABE K G, RUBIN J, et al. Pancreatic neuroendocrine tumors (PNETs): Incidence, prognosis and recent trend toward improved survival[J]. Ann Oncol, 2008, 19(10): 1727–1733.
[2] BILIMORIA K Y, TALAMONTI M S, TOMLINSON J S, et al. Prognostic score predicting survival after resection of pancreatic neuroendocrine tumors: Analysis of 3851 patients[J]. Ann Surg, 2008, 247(3): 490–500.
[3] SCOAZEC J Y, COUVELARD A. Classification of pancreatic neuroendocrine tumours: Changes made in the 2017 WHO classification of tumours of endocrine organs and perspectives for the future[J]. Ann Pathol, 2017, 37(6): 444–456.
[4] METZ D C, JENSEN R T. Gastrointestinal neuroendocrine tumors: pancreatic endocrine tumors[J]. Gastroenterology, 2008, 135(5): 1469–1492.
[5] DASARI A, SHEN C, HALPERIN D, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States[J]. JAMA Oncol, 2017, 3(10): 1335–1342.
[6] WEN J, CHEN J, LIU D, et al. The eighth edition of the American Joint Committee on Cancer distant metastases stage classification for metastatic pancreatic neuroendocrine tumors might be feasible for metastatic pancreatic ductal adenocarcinomas[J]. Neuroendocrinology, 2020, 110(5): 364–376.
[7] LUO G, JAVED A, STROSBERG J R, et al. Modified staging classification for pancreatic neuroendocrine tumors on the basis of the American Joint Committee on Cancer and European Neuroendocrine Tumor Society Systems[J]. J Clin Oncol, 2017, 35(3): 274–280.
[8] JILESEN A P, VAN EIJCK C H, IN'T HOF K H, et al. Postoperative complications, in-hospital mortality and 5-year survival after surgical resection for patients with a pancreatic neuroendocrine tumor: A systematic review[J]. World J Surg, 2016, 40(3): 729–748.
[9] GAO L, NATOV N S, DALY K P, et al. An update on the management of pancreatic neuroendocrine tumors[J]. Anticancer Drugs, 2018, 29(7): 597–612.
[10] ORDITURA M, PETRILLO A, VENTRIGLIA J, et al. Pancreatic neuroendocrine tumors: Nosography, management and treatment[J]. Int J Surg, 2016, 28 Suppl 1: S156–S162.
[11] BAUR A D, PAVEL M, PRASAD V, et al. Diagnostic imaging of pancreatic neuroendocrine neoplasms (pNEN): Tumor detection, staging, prognosis, and response to treatment[J]. Acta Radiol, 2016, 57(3): 260–270.
[12] WANG Z X, QIU M Z, JIANG Y M, et al. Comparison of prognostic nomograms based on different nodal staging systems in patients with resected gastric cancer[J]. J Cancer, 2017, 8(6): 950–958.
[13] OHMOTO A, ROKUTAN H, YACHIDA S. Pancreatic neuroendocrine neoplasms: Basic biology, current treatment strategies and prospects for the future[J]. Int J Mol Sci, 2017, 18(1): 143.
[14] FANG C, WANG W, FENG X, et al. Nomogram individually predicts the overall survival of patients with gastroenteropancreatic neuroendocrine neoplasms[J]. Br J Cancer, 2017, 117(10): 1544–1550.
[15] MILIONE M, MICELI R, BARRETTA F, et al. Microenvironment and tumor inflammatory features improve prognostic prediction in gastro-entero-pancreatic neuroendocrine neoplasms[J]. J Pathol Clin Res, 2019, 5(4): 217–226.
[16] SCOTT A T, HOWE J R. Evaluation and management of neuroendocrine tumors of the pancreas[J]. Surg Clin North Am, 2019, 99(4): 793–814.
[17] BETTINI R, PARTELLI S, BONINSEGNA L, et al. Tumor size correlates with malignancy in nonfunctioning pancreatic endocrine tumor[J]. Surgery, 2011, 150(1): 75–82.
[18] GAUJOUX S, PARTELLI S, MAIRE F, et al. Observational study of natural history of small sporadic nonfunctioning pancreatic neuroendocrine tumors[J]. J Clin Endocrinol Metab, 2013, 98(12): 4784–4789.
[19] FINKELSTEIN P, SHARMA R, PICADO O, et al. Pancreatic neuroendocrine tumors (panNETs): Analysis of overall survival of nonsurgical management versus surgical resection[J]. J Gastrointest Surg, 2017, 21(5): 855–866.
[20] MEI W, DING Y, WANG S, et al. Head and body/tail pancreatic neuroendocrine tumors have different biological characteristics and clinical outcomes[J]. J Cancer Res Clin Oncol, 2020, 146(11): 3049–3061.
[21] LIU M, SUN X, ZHANG Z, et al. The clinical characteristics and survival associations of pancreatic neuroendocrine tumors: Does age matter?[J]. Gland Surg, 2021, 10(2): 574–583.
[22] ZHOU H, ZHANG Y, SONG Y, et al. Marital status is an independent prognostic factor for pancreatic neuroendocrine tumors patients: An analysis of the Surveillance, Epidemiology, and End Results (SEER) database[J]. Clin Res Hepatol Gastroenterol, 2017, 41(4): 476–486.
Prognostic prediction model construction and validation of pancreatic neuroendocrine tumor
Department of Digestive, Linyi Central Hospital, Linyi 276400, Shandong, China
To investigate the risk factors affecting the prognosis of pancreatic neuroendocrine tumors, and to construct a prognosis prediction model of pancreatic neuroendocrine tumors.A total of 3606 patients diagnosed with pancreatic neuroendocrine tumor from January 2004 to December 2015 were selected from the SEER database. They were divided into training set (=2704) and validation set (=902) according to the ratio of 3:1. In training set, the Cox proportional hazards model was used to screen the effects of pancreatic neuroendocrine tumors prognostic risk factors, further construct its prognostic model and draw nomogram. The predictive performance of the model was validated internally and externally on the training set and validation set, respectively.Univariate Cox regression analysis showed that gender, age, marital status, tumor location, degree of differentiation, TNM stage, American Joint Committee on Cancer (AJCC) stage, and surgery or not were risk factors for the prognosis of pancreatic neuroendocrine tumors (<0.05). Multivariate Cox regression analysis showed that age, gender, marital status, degree of differentiation, TNM stage and surgery were the risk factors affecting the prognosis of pancreatic neuroendocrine tumors (<0.05). Finally, variables such as age, gender, degree of differentiation, tumor location, TNM stage, surgery, and marital status were incorporated into the prediction model and nomograms were drawn to predict 3-year and 5-year survival rates. In training set and validation set, the C-index of the prediction model was 0.8579 and 0.8572, respectively. The calibration curves of the 3-year and 5-year survival rates in the training set and the verification set showed that the predicted survival rate was in good agreement with the actual survival rate.The constructed pancreatic neuroendocrine tumor prediction model has good predictive value.
Pancreatic neuroendocrine tumor; Prediction model; SEER database
R735.9
A
10.3969/j.issn.1673-9701.2023.07.014
陳慶法,電子信箱:qingfachen@163.com
(2022–09–05)
(2023–02–06)

