松齡血脈康膠囊對大鼠腦缺血再灌注損傷 內質網應激凋亡相關蛋白的影響
馬喆 王思錦 高永紅 程煒婷 鞏卓彥 孫逸坤 薛程元 金秋碩 高穎 婁利霞
〔摘要〕 目的 探討松齡血脈康膠囊對大鼠腦缺血再灌注損傷的保護作用及其對內質網應激細胞凋亡的影響。方法 從50只雄性SD大鼠中隨機選取11只為假手術組,其余大鼠采用大腦中動脈栓塞法制備腦缺血再灌注損傷模型,造模成功的33只大鼠隨機分為模型組、松齡血脈康組及依達拉奉組,每組11只,分別予蒸餾水[10 mL/(kg·d)]、松齡血脈康膠囊溶液[10 mL/(kg·d)]及依達拉奉注射液[3.15 mL/(kg·d)]干預5 d。神經功能評分檢測造模后24 h及給藥5 d后的神經功能;錯步實驗檢測錯步次數;HE染色觀察大鼠大腦缺血區形態結構;TUNEL染色法觀察細胞凋亡;Western blot法檢測Cleaved Caspase-3及內質網應激促凋亡蛋白CHOP、Caspase-12的表達。結果 神經功能評分顯示,造模24 h后,與假手術組相比,造模組評分顯著降低(P<0.01),表示造模成功。造模5 d后,模型組神經功能評分仍低于假手術組(P<0.01);松齡血脈康組與依達拉奉組神經功能評分顯著高于模型組(P<0.05),且分別顯著高于本組造模后24 h評分(P<0.01或P<0.05)。錯步實驗檢測發現,模型組的錯步次數顯著高于假手術組(P<0.01);松齡血脈康組及依達拉奉組錯步次數顯著低于模型組(P<0.01)。HE染色結果發現,與模型組相比,松齡血脈康組及依達拉奉組缺血區腦組織形態結構顯著改善。TUNEL染色結果顯示,模型組陽性細胞百分比較假手術組顯著增加(P<0.01);松齡血脈康組及依達拉奉組陽性細胞百分比較模型組顯著減少(P<0.01)。Western blot檢測結果顯示,模型組Cleaved Caspase-3與CHOP、Caspase-12相對蛋白表達量較假手術組顯著增高(P<0.01);松齡血脈康組及依達拉奉組Cleaved Caspase-3及CHOP、Caspase-12相對蛋白表達量較模型組明顯減少(P<0.05或P<0.05)。結論 松齡血脈康膠囊可能是通過降低內質網應激促凋亡蛋白CHOP、Caspase-12的表達減少細胞凋亡,減輕腦缺血再灌注損傷,發揮神經保護作用。
〔關鍵詞〕 腦缺血再灌注損傷;松齡血脈康膠囊;依達拉奉注射液;內質網應激;細胞凋亡;凋亡蛋白
〔中圖分類號〕R255.2? ? ? ? 〔文獻標志碼〕A? ? ? ?〔文章編號〕doi:10.3969/j.issn.1674-070X.2021.05.006
Effects of Songling Xuemaikang Capsule on Endoplasmic Reticulum Stress Related Apoptosis Proteins in Rats with Cerebral Ischemia Reperfusion Injury
MA Zhe1, WANG Sijin1,2, GAO Yonghong1, CHENG Weiting1, GONG Zhuoyan1, SUN Yikun1, XUE Chengyuan1,
JIN Qiushuo1, GAO Ying1,2, LOU Lixia1*
(1. Ministry of Education and Beijing Key Laboratory of Chinese Internal Medicine, Dongzhimen Hospital, Beijing
University of Chinese Medicine, Beijing 100700, China; 2. Institute of Traditional Chinese Encephalopathy, Beijing
University of Chinese Medicine, Beijing 100700, China)
〔Abstract〕 Objective To investigate the protective effect of Songling Xuemaikang Capsule on cerebral ischemia reperfusion injury in rats and its effect on the apoptosis of endoplasmic reticulum stress cells. Methods A total of 11 male SD rats were randomly selected from 50 male SD rats as the sham group. The other rats were established by right middle cerebral artery embolization to prepare cerebral ischemia reperfusion injury model. 33 rats were successfully modeled, and were randomly divided into model group, Songling Xuemaikang group and edaravone group, with 11 rats in each group. And they were respectively given distilled water [10 mL/(kg·d)], Songling Xuemaikang Capsules solution [10 mL/(kg·d)] and edaravone injection [3.15 mL/(kg·d)]. They were all treated for 5 days. Neurological function score was used to detect the neurological function 24 hours after modeling and 5 days after administration. Stagger-step experiment was used to detect the number of stagger-step. HE staining was used to observe the morphology and structure of cerebral ischemia in rats. Cell apoptosis was observed by TUNEL staining. The expression of Cleaved Caspase-3 and endoplasmic reticulum stress pro-apoptotic proteins CHOP and Caspase-12 were detected by Western blot. Results Neurological function score showed that, 24 hours after modeling, the score of the modeling group was significantly lower than that of the sham group (P<0.01), indicating that the modeling was successful. After 5 days of modeling, the neurological function score of model group was still lower than that of sham group (P<0.01). The neurological function score in Songling Xuemaikang group and edaravone group was significantly higher than that in model group (P<0.05), and was significantly higher than that in self group 24 hours after modeling (P<0.05, P<0.01) respectively. Stagger-step experiment showed the number of wrong steps in the model group was significantly higher than that in the sham group (P<0.01). The number of wrong steps in Songling Xuemaikang group and edaravone group was significantly lower than that in model group (P<0.01). The results of HE staining showed that, compared with the model group, the morphological structure of ischemic brain tissue in Songling Xuemaikang group and edaravone group was significantly improved. TUNEL staining showed that the percentage of positive cells in the model group was significantly higher than that in the sham group (P<0.01). The percentage of positive cells in Songling Xuemaikang group and edaravone group was significantly lower than that in model group (P<0.01). Western blot results showed that the relative protein expression levels of Cleaved Caspase-3, CHOP and Caspase-12 in model group were significantly higher than those in sham group (P<0.01). The relative protein expression levels of Cleaved Caspase-3, CHOP and Caspase-12 in Songling Xuemaikang group and edaravone group were significantly decreased compared with model group (P<0.05 or P<0.01). Conclusion Songling Xuemaikang Capsule may reduce cell apoptosis by reducing the expression of endoplasmic reticulum stress pro-apoptotic proteins CHOP and Caspase-12, alleviate cerebral ischemia reperfusion injury and play a neuroprotective role.
1.8? TUNEL染色法檢測大鼠腦組織細胞凋亡
根據試劑盒操作說明,將大鼠腦組織石蠟切片脫蠟、水化,組織固定于室溫30 min,用PBS進行清洗,滴加蛋白酶K工作液(100 μL/片)室溫孵育10 min進行消化,PBS清洗后再次室溫固定5 min。PBS清洗后,滴加Equilibration Buffer液(100 μL/片)室溫孵育10 min用于反應前平衡,吸去Equilibration Buffer液,滴加含熒光標記底物的末端DNA轉移酶反應混合液(50 μL/片),37 ℃避光孵育60 min,2×SSC溶液清洗,最后滴加內含DAPI成分的抗熒光衰減封片劑(10 μL/片)封片。在熒光顯微鏡下觀測,每張切片隨機選擇10個腦組織缺血區,在400倍鏡視野下進行拍照,用Image J 軟件對凋亡細胞數量進行統計,細胞凋亡率=陽性細胞數/總細胞數×100%。
1.9? Western blot 法檢測各組大鼠腦組織的Cleaved Caspase-3、CHOP、Caspase-12蛋白表達
取大鼠缺血區腦組織50 mg,加入其10倍體積的裂解液進行蛋白提取,蛋白濃度的測定采用BCA法,并將其濃度全部調整為4 μg/μL,分裝凍存于
-80 ℃冰箱備用。采用快速凝膠法制備12.5%的PAGE膠,電泳將不同分子量的蛋白分離(上樣量:40 μg),并將其轉移至孔徑為0.22 μm的NC膜,采用Western封閉液室溫封閉1 h后分別加入一抗Clea?
ved Caspase-3、CHOP、Caspase-12(比例均為1∶1 000)及GAPDH(1∶10 000)、β-actin(1∶20 000),將加入一抗的NC膜存放于4 ℃冰箱孵育過夜,次日回收一抗,清洗后加入羊抗兔二抗(1∶20 000)室溫孵育1 h,洗滌后在暗環境中滴加發光液并使用凝膠成像儀記錄,Image J 軟件分析條帶相對灰度值,β-actin或GAPDH為內參。
1.10? 統計學方法
實驗數據通過SPSS 20.0軟件進行統計分析,數據結果用“x±s”表示。符合正態分布且方差齊的數據,采用單因素方差分析,組間兩兩比較采取LSD檢驗進行;符合正態分布但方差不齊的數據,組間比較則采用單因素方差分析中Dunnett T3檢驗。P<0.05表示差異具有統計學意義。
2 結果
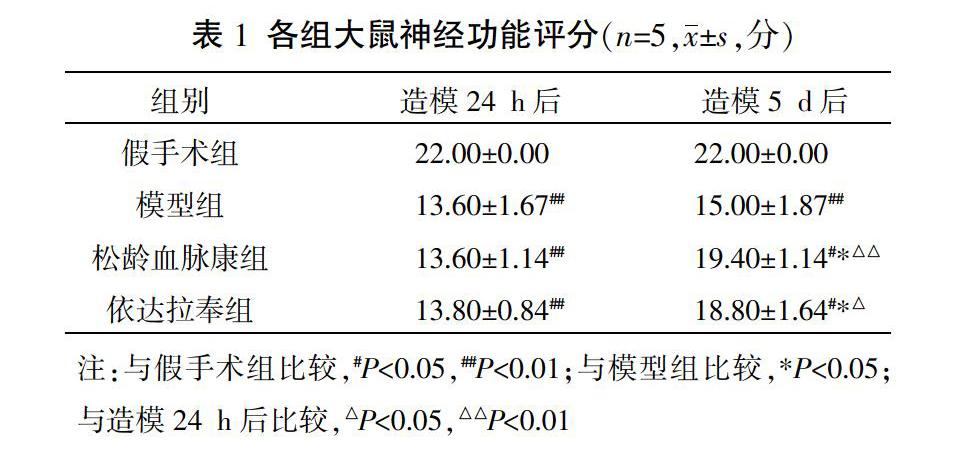
2.1? 各組大鼠神經功能評分比較
造模24 h后,假手術組神經功能評分為22分,模型組、松齡血脈康組及依達拉奉組評分均低于假手術組,差異有統計學意義(P<0.01),表示造模成功。造模5 d后,模型組、松齡血脈康組與依達拉奉組神經功能評分仍低于假手術組,差異有統計學意義(P<0.01,P<0.05);松齡血脈康組與依達拉奉組神經功能評分顯著高于模型組,差異有統計學意義(P<0.05)。與造模24 h后比較,假手術組及模型組在造模5 d后的評分無顯著差異(P>0.05);松齡血脈康組及依達拉奉組神經功能評分顯著高于造模24 h后評分,差異有統計學意義(P<0.01,P<0.05)。見表1。
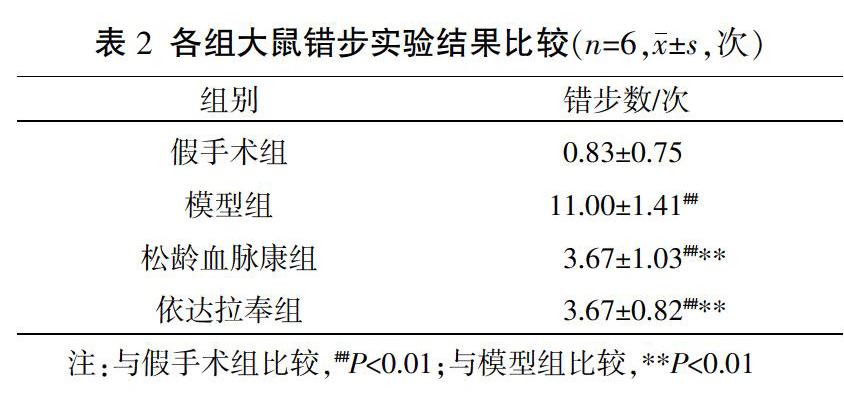
2.2? 各組大鼠錯步實驗結果比較
與假手術組比較,模型組、松齡血脈康組及依達拉奉組大鼠的錯步次數顯著增加,差異有統計學意義(P<0.01);與模型組比較,松齡血脈康組及依達拉奉組大鼠錯步次數顯著減少,差異有統計學意義(P<0.01);松齡血脈康組與依達拉奉組相比,差異無統計學意義(P>0.05)。見表2。
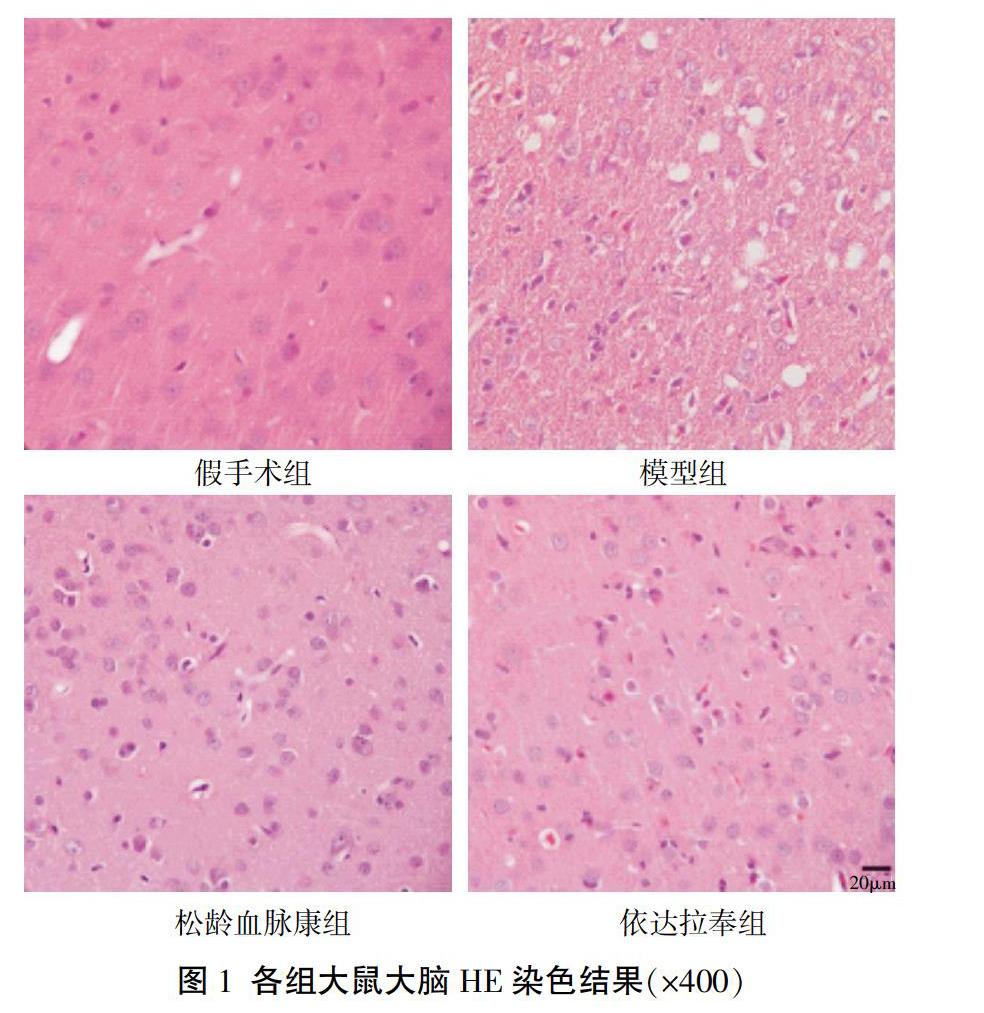
2.3? HE染色觀察各組大鼠腦組織形態結構改變
假手術組大腦皮層神經元細胞形態完整、結構正常、胞核清晰,胞質充盈,細胞排列整齊;模型組大腦皮層神經元細胞皺縮,染色質色深濃,胞核與胞質無明顯分界,部分細胞呈空泡狀改變;與模型組比較,松齡血脈康組及依達拉奉組大腦皮層神經元細胞皺縮減少、細胞形態及結構明顯改善。見圖1。
2.4? TUNEL染色觀察各組大鼠腦組織細胞凋亡情況
假手術組無陽性細胞染色;與假手術組相比,模型組、松齡血脈康組及依達拉奉組陽性細胞百分比顯著增加,差異有統計學意義(P<0.01);與模型組相比,松齡血脈康組及依達拉奉組陽性細胞百分比顯著減少,差異有統計學意義(P<0.01);與依達拉奉組相比,松齡血脈康組陽性細胞百分比顯著減少,差異有統計學意義(P<0.01)。見圖2、表3。
2.5? 各組大鼠腦組織調亡蛋白Cleaved Caspase-3及ERS促凋亡蛋白CHOP和 Caspase-12的表達
與假手術組相比,模型組、松齡血脈康組及依達拉奉組Cleaved Caspase-3與CHOP、Caspase-12相對蛋白表達量顯著增高,差異有統計學意義(P<0.01);與模型組相比,松齡血脈康組及依達拉奉組Cleaved Caspase-3及CHOP、Caspase-12相對蛋白表達量明顯減少,差異有統計學意義(P<0.05或P<0.01);與依達拉奉組相比,松齡血脈康組Cleaved Caspase-3及CHOP、Caspase-12相對蛋白表達差異無統計學意義(P>0.05)。見圖3、表4。
3 討論
松齡血脈康膠囊成分為鮮松葉、珍珠層粉、葛根;鮮松葉具有祛風燥濕、活血安神等作用。《本草匯言》記載松毛能治“大風癩疾”;珍珠層粉具有重鎮平肝、清熱涼血等作用,《本草匯言》記錄珍珠層粉“鎮心,定志”;葛根具有解肌生津、升陽止瀉等作用,是陽明經引經藥,《本草正》記錄葛根“凡熱而兼渴者,此為最良”;三藥相伍,共奏平肝潛陽息風、鎮心安神之效。CIRI的中醫病機為肝陽上亢,氣血逆亂,上犯于腦,因此,從中醫學理論角度而言,松齡血脈康可以治療CIRI。目前的研究[10-12]表明,鮮松葉、葛根及珍珠層粉均具有抗氧化應激的作用。CIRI時會產生大量活性氧(reactive oxygen species, ROS),發生脂質過氧化反應,破壞內質網的Ca2+穩態,從而誘發ERS介導的細胞凋亡[13]。因此,推測松齡血脈康可以通過減輕ERS改善CIRI。依達拉奉注射液具有抗ERS凋亡、清除氧自由基的作用,在CIRI中發揮神經保護作用[14],故作為本研究的陽性對照組。
缺血性腦卒中急性期血管再通后,腦組織缺血區的半暗帶受到再灌注損傷,而細胞凋亡是CIRI的重要機制之一[15-16]。CIRI的治療旨在減少細胞凋亡,減輕組織灌注不足帶來的影響,挽救缺血半暗帶,促進神經功能恢復等[17]。在本研究中,造模24 h后模型組大鼠神經功能評分較低(P<0.01),且與造模5 d后相比,其神經功能無顯著差異;松齡血脈康組與依達拉奉組在干預5 d后神經功能評分顯著高于模型組(P<0.05),并且分別高于本組造模24 h后評分(P<0.01,P<0.05),同時,松齡血脈康組與依達拉奉組神經功能評分無顯著差異。表明松齡血脈康膠囊及依達拉奉注射液可以改善CIRI后大鼠的神經功能,并且松齡血脈康膠囊和依達拉奉注射液的效果相當。錯步實驗可以檢測大鼠共濟失調情況,模型組大鼠錯步次數顯著增加(P<0.01),經過松齡血脈康膠囊與依達拉奉注射液治療后其錯步次數顯著減少(P<0.01),說明松齡血脈康膠囊可以改善CIRI大鼠共濟失調,并且和依達拉奉注射液效果相當。HE染色中,經過松齡血脈康膠囊和依達拉奉注射液治療后神經元細胞形態結構較模型組改善。以上研究表明松齡血脈康膠囊可以改善大鼠CIRI缺血區神經元細胞形態結構,具有神經保護作用。
已有研究[18]表明,ERS是CIRI細胞凋亡信號通路的關鍵組成部分。內質網是蛋白質合成的重要場所,并且參與鈣離子穩態調節、脂質和葡萄糖代謝[19]。當CIRI發生時,內質網的穩態被破壞,產生ERS,導致蛋白質合成障礙而使錯誤折疊蛋白產生并積聚,從而激活未折疊蛋白反應(unfolded protein response, UPR),最初UPR的激活可以幫助重建內質網穩態并使內質網功能恢復正常,但是持久而嚴重的CIRI激活ERS凋亡通路發生細胞凋亡[20]。CHOP是ERS信號通路下游重要的促凋亡因子,參與內質網介導的細胞凋亡[21]。現有研究[22]表明CHOP基因沉默可以減輕ERS誘導的細胞凋亡。CHOP蛋白的表達可通過ERS信號通路PERK-elF2-ATF4及IRE1a-JNK調節,ERS激活PERK及IRE1a,增加CHOP蛋白的表達,調節Bcl-2蛋白及Bax蛋白的表達并且啟動Caspase凋亡信號通路發生細胞凋亡[23]。Caspase-12是ERS凋亡通路中另一個重要的蛋白,存在于內質網膜中,通常以無活性的前體形式存在。已有的研究[24]證明ERS介導的細胞凋亡可以通過抑制 Caspase-12蛋白表達而減輕。ERS可以激活IRE1,活化的IRE1激活Caspase-12,啟動內質網介導的細胞凋亡[25]。Caspase-12活化并裂解Caspase-9,后者進一步活化裂解Caspase-3,最終導致細胞凋亡。在本研究中,模型組Cleaved Caspase-3與CHOP、Caspase-12相對蛋白表達量顯著增高(P<0.01),而經松齡血脈康膠囊及依達拉奉注射液干預5 d后,其蛋白表達量顯著減少(P<0.05或P<0.01)。并且TUNEL染色顯示模型組陽性細胞百分比顯著增加(P<0.01),而經松齡血脈康膠囊及依達拉奉注射液干預5 d后其陽性細胞百分比顯著減少(P<0.01),且松齡血脈康組比依達拉奉組細胞凋亡百分比低(P<0.01)。以上結果表明松齡血脈康膠囊可以減少ERS引起的細胞凋亡,并且其對細胞凋亡的作用與依達拉奉注射液相仿。因此,推測松齡血脈康膠囊可能是通過降低ERS特異的促凋亡蛋白CHOP、Caspase-12的表達水平減少細胞凋亡,減輕CIRI,發揮神經保護作用。
參考文獻
[1] ROGER V L, GO A S, LLOYD-JONES D M, et al. Executive summary: Heart disease and stroke statistics: 2012 update: A report from the American heart association[J]. Circulation, 2012, 125(1): 188-97.
[2] 王擁軍,李子孝,谷鴻秋,等.中國卒中報告2019(中文版)(1)[J].中國卒中雜志,2020,15(10):1037-1043.
[3] 蔡周權,袁浩宇,俞? 瑜.松齡血脈康膠囊聯合辛伐他汀治療心絞痛伴高脂血癥48例臨床評價[J].中國藥業,2015,24(17):29-30.
[4] 梁汝慶,姜? 婧,丁新生,等.松齡血脈康對大鼠局灶性腦缺血-再灌注損傷和細胞凋亡的影響[J].中國新藥與臨床雜志,2010,29(3): 221-224.
[5] 陳? 媛,吳海金,黃曉松,等.葛根素對腦缺血再灌注大鼠海馬組織P-STAT3、P53表達的影響[J].湖南中醫藥大學學報,2018,38(1): 36-39.
[6] 李春紅,王? 婕,吳愛明,等.內質網應激與心力衰竭的相關性研究進展[J].中西醫結合心腦血管病雜志,2018,16(12):1662-1666.
[7] LONGA E Z, WEINSTEIN P R, CARLSON S, et al. Reversible middle cerebral artery occlusion without craniectomy in rats[J]. Stroke, 1989, 20(1): 84-91.
[8] GARCIA J H, WAGNER S, LIU K F, et al. Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats. Statistical validation[J]. Stroke, 1995, 26(4): 627-634,635.
[9] METZ G A, WHISHAW I Q. Cortical and subcortical lesions impair skilled walking in the ladder rung walking test: A new task to evaluate fore-and hindlimb stepping, placing, and co-ordination[J]. Journal of Neuroscience Methods, 2002, 115(2): 169-179.
[10] 勞業興,張冰若,蘇薇薇.松針化學成分及藥理研究進展[J].中藥材,2003,26(9):681-683.
[11] 李? 端,徐? 翔,吳佩君,等.水解珍珠層粉在小鼠體內的抗氧化作用[J].中成藥,1996(12):30-31.
[12] 毛慶軍,夏? 瑞,張傳漢.葛根素對腦缺血再灌注家兔腦組織及內皮細胞的保護[J].中國臨床康復,2006(3):40-41.
[13] 周映彤,肖洪彬,畢明剛.活性氧與內質網應激[J].中國藥理學通報,2011,27(5):597-600.
[14] KIKUCHI K, TANCHAROEN S, TAKESHIGE N, et al. The efficacy of edaravone (radicut), a free radical scavenger, for cardiovascular disease[J]. International Journal of Molecular Sciences, 2013, 14(7): 13909-13930.
[15] MANDALANENI K ,RAYI A, JILLELLA D V. Stroke reperfusion injury[M]. Internet: StatPearls, 2020: 1-24.
[16] 雷夢南,李? 玉,胡建鵬.腦缺血再灌注損傷分子生物學機制及現代中醫藥治療進展[J].長春中醫藥大學學報,2019,35(5):991-994.
[17] SAVITZ S I, BARON J C, YENARI M A, et al. Reconsidering neuroprotection in the reperfusion era[J]. Stroke, 2017, 48(12): 3413-3419.
[18] 李欲軻,熊孟連,徐? 僡,等.內質網應激與凋亡研究進展[J].分子植物育種,2018,16(23):7856-7862.
[19] PRENTICE H, MODI J P, WU J Y. Mechanisms of neuronal protection against excitotoxicity, endoplasmic Reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative
diseases[J]. Oxidative Medicine and Cellular Longevity, 2015, 2015: 964518.
[20] LALKOVIOV M, DANIELISOV V. Neuroprotection and antioxidants[J]. Neural Regeneration Research, 2016, 11(6):865-74.
[21] WALTER P, RON D. The unfolded protein response: From stress pathway to homeostatic regulation[J]. Science, 2011, 334(6059): 1081-1086.
[22] 雷? 艷,詹世淮,陳俊秋,等.CHOP雙重調控衣霉素誘導的DU-145細胞凋亡及自噬的研究[J].中華細胞與干細胞雜志(電子版),2018,8(5):257-263.
[23] LI Y M, GUO Y S, TANG J, et al. New insights into the roles of CHOP-induced apoptosis in ER stress[J]. Acta Biochimica et Biophysica Sinica, 2014, 46(8): 629-640.
[24] KIKUCHI K, TANCHAROEN S, TAKESHIGE N, et al. The efficacy of edaravone (radicut), a free radical scavenger, for cardiovascular disease[J]. International Journal of Molecular Sciences, 2013, 14(7): 13909-13930.
[25] ZHAO X M, ZHU L, LIU D Y, et al. Sigma-1 receptor protects against endoplasmic Reticulum stress-mediated apoptosis in mice with cerebral ischemia/reperfusion injury[J]. Apoptosis, 2019, 24(1/2): 157-167.

